Advertisements
Advertisements
प्रश्न
Name the radiation of the electromagnetic spectrum which is used for the following:
To photograph internal parts of the human body and Give the frequency range
उत्तर
X-Rays (1016 to 1019)Hz
APPEARS IN
संबंधित प्रश्न
Name the parts of the electromagnetic spectrum which is
used to treat muscular strain.
Write in brief, how these waves can be produced.
What physical quantity is the same for X-rays of wavelength 10−10 m, red light of wavelength 6800 Å and radiowaves of wavelength 500 m?
The small ozone layer on top of the stratosphere is crucial for human survival. Why?
If the earth did not have an atmosphere, would its average surface temperature be higher or lower than what it is now?
(a) Give a list of at least five radiations, in order of their increasing frequencies, which make up the complete electromagnetic spectrum.
(b) Which of the radiation mentioned by you in part (a) has the highest penetrating power.
An electromagnetic wave has a frequency of 500 MHz and a wavelength of 60 cm Calculate the velocity of the wave.
Consider a photon of a continuous X-ray coming from a Coolidge tube. Its energy comes from
When 40 kV is applied across an X-ray tube, X-ray is obtained with a maximum frequency of 9.7 × 1018 Hz. Calculate the value of Planck constant from these data.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Answer briefly.
Name the most harmful radiation entering the Earth's atmosphere from outer space.
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
The figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00eV, E2 = 1.78eV and E3 = 2.95eV. The ground state is considered 0 eV for reference. The transition of electrons takes place between levels E1 and E2.
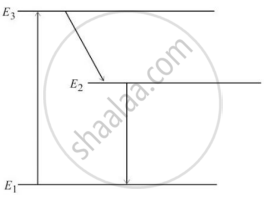
- What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
- Suppose the external source has a power of 100 W. What would be the rate of photon emission?
