Advertisements
Advertisements
प्रश्न
Pragya tested the solubility of three different substances at different temperatures and collected the data as given below (results are given in the following table, as grams of substance dissolved in 100 grams of water to form a saturated solution).
| Substance dissolved | Temperature in K | ||||
| 283 | 293 | 313 | 333 | 353 | |
| Solubility | |||||
| Potassium nitrate | 21 | 32 | 62 | 106 | 167 |
| Sodium chloride | 36 | 36 | 36 | 37 | 37 |
| Potassium chloride | 35 | 35 | 40 | 46 | 54 |
| Ammonium chloride | 24 | 37 | 41 | 55 | 66 |
- What mass of potassium nitrate would be needed to produce a saturated solution of potassium nitrate in 50 grams of water at 313 K?
- Pragya makes a saturated solution of potassium chloride in water at 353 K and leaves the solution to cool at room temperature. What would she observe as the solution cools? Explain.
- Find the solubility of each salt at 293 K. What salt has the highest solubility at this temperature?
- What is the effect of change of temperature on the solubility of a salt?
उत्तर
a) Solubility of potassium nitrate at 313 K `62/100`
100 g of water contains potassium nitrate = 62g
50 g of water contains potassium nitrate = `62/100 xx 50` = 31 g
Thus, 31 g of potassium nitrate would be needed to produce a saturated solution of potassium nitrate in 50 g of water at 313K
b) At 313 K, dissolve magnesium nitrate in 50 g of water. (a) As a saturated solution of potassium chloride at 353 K is cooled, its solubility in water drops. As a result, crystals form when the amount of potassium chloride exceeds its solubility at lower temperatures.
c) Solubility refers to the quantity of salt that may dissolve in 100 g of water to create a saturated solution at a specific temperature.
Solubility of potassium nitrate at 293 K = `32/100`
Solubility of sodium chloride at 293 K = `36/100`
Solubility of potassium chloride at 293 K = `35/100`
Solubility of ammonium chloride at 293 K = `37/100`
From the above data, it is clear that ammonium chloride has the highest solubility at 293 K.
d) In general, a salt's solubility increases with temperature. However, the rise varies depending on the salt. For example, the solubility of potassium nitrate increases significantly, that of ammonium chloride increases somewhat, that of potassium chloride increases marginally, and that of sodium chloride remains nearly unchanged.
APPEARS IN
संबंधित प्रश्न
The number of components in a binary solution is ______
Define the term: Solution
Give an example of gas in a liquid.
Give an example of solid in solid.
The teacher instructed three students ‘A’, ‘B’ and ‘C’ respectively to prepare a 50% (mass by volume) solution of sodium hydroxide (NaOH). ‘A’ dissolved 50g of NaOH in 100 mL of water, ‘B’ dissolved 50g of NaOH in 100g of water while ‘C’ dissolved 50g of NaOH in water to make 100 mL of solution. Which one of them has made the desired solution and why?
A solution is always a liquid. Comment.
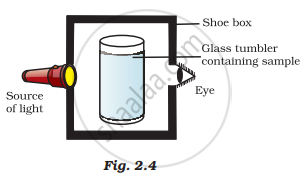
A group of students took an old shoebox and covered it with black paper from all sides. They fixed a source of light (a torch) at one end of the box by making a hole in it and making another hole on the other side to view the light. They placed a milk sample contained in a beaker/tumbler in the box as shown in Fig.2.4. They were amazed to see that milk taken in the tumbler was illuminated. They tried the same activity by taking a salt solution but found that light simply passed through it?
(a) Explain why the milk sample was illuminated. Name the phenomenon involved.
(b) The same results were not observed with a salt solution. Explain.
(c) Can you suggest two more solutions that would show the same effect as shown by the milk solution?
During an experiment, the students were asked to prepare a 10% (Mass/Mass) solution of sugar in water. Ramesh dissolved 10g of sugar in 100g of water while Sarika prepared it by dissolving 10g of sugar in water to make 100g of the solution.
(a) Are the two solutions of the same concentration
(b) Compare the mass % of the two solutions.
When the solid (Solute) dissolves in a liquid (Solvent), it becomes a solution.
______ is the major difference between true solutions, suspensions, and colloids.
