Advertisements
Advertisements
प्रश्न
Silver articles become black on prolonged exposure to air. This is due to the formation of
पर्याय
Ag3N
Ag2O
Ag2S
Ag2S and Ag3N
उत्तर
Ag2S
Explanation -
Silver objects corrode slowly in the air due to the presence of hydrogen sulphide which reacts with silver articles to form a black coloured chemical compound silver sulphide.
`4"Ag"("s") + 2"H"_2"S"("g") + "O"_2("g") -> 2"Ag"_2"S"("s") + 2"H"_2"O"("l")`
APPEARS IN
संबंधित प्रश्न
Give two examples of amphoteric oxides.
Give reason:
Sodium, potassium and lithium are stored under oil.
Which of the following oxide(s) of iron would be obtained on the prolonged reaction of iron with steam?
Example of an amphoteric oxide is:
Which one of the following metals does not react with cold as well as hot water?
Which of the following oxide(s) of iron would be obtained on prolonged reaction of iron with steam?
Answer the questions based on the figure below:
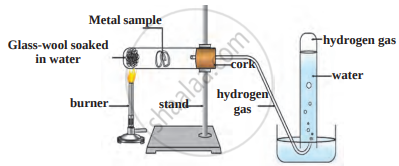
- Which reaction is shown in the figure?
- Which gas is evolved in the reaction?
- Give an example of reactants that rapidly show this reaction. Give equations.
- Give an example of reactants that do not react rapidly.
- In what condition will reactants of (c) part react? Give equation.
Keerti added dilute Hydrochloric acid to four metals and recorded her observations as shown in the table given below:
| Metal | Gas Evolved |
| Copper | Yes |
| Iron | Yes |
| Magnesium | No |
| Zinc | Yes |
Select the correct observation(s) and give chemical equation(s) of the reaction involved.
Metal oxides generally react with acids, but few oxides of metal also react with bases. Such metallic oxides are:
- MgO
- ZnO
- Al2O3
- Cao
Select the appropriate state symbols of the products given as X and Y in the following chemical equation by choosing the correct option from table given below:
\[\ce{Zn_{(s)} + H_2SO_{4(l)} ->ZnSO_{4(X)} + H_{2(Y)}}\]
