Advertisements
Advertisements
प्रश्न
Specify the oxidation number of the metal in the following coordination entity:
[CoBr2(en)2]+
उत्तर
Let the oxidation number of metal be x.
Hence, x + 2 × (−1) + 2 × (0) = +1
x = 1 + 2
∴ x = +3
APPEARS IN
संबंधित प्रश्न
Write the IUPAC name of 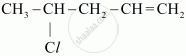
IUPAC name of K4[Fe(CN)6] is
Write the IUPAC name of the given compound:
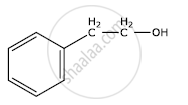
Write the IUPAC name of the given compound:
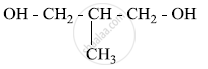
Write the IUPAC name of the given compound:
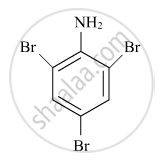
Write down the IUPAC name of the complex [Pt(en)2Cl2]2+. What type of isomerism is shown by this complex?
Write the IUPAC name of the complex [Cr(NH3)4 Cl2]Cl.
Write the IUPAC name of the following coordination compound:
K2[PdCl4]
Specify the oxidation number of the metal in the following coordination entity:
[PtCl4]2−
Using IUPAC norms write the systematic name of the following:
[Pt(NH3)2Cl(NH2CH3)]Cl
Using IUPAC norms, write the systematic name of the following:
[NiCl4]2−
Using IUPAC norms, write the systematic name of the following:
[Co(en)3]3+
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
[Co(NH3)5Cl]Cl2
The oxidation number of cobalt in K[Co(CO)4] is
(i) +1
(ii) +3
(iii) −1
(iv) −3
Write down the IUPAC name of the following complex :
[Cr(en)3]Cl3
Write the IUPAC name of the compound
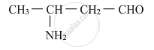
Name the type of isomerism shown by the following compounds:
[CU(NH3)4] [PtCl4] and [Pt(NH3)4] [CuCl4]
Write the formula for the following coordination compound.
Bis (ethane-1,2-diamine) dihydroxidochromium (III) chloride
