Advertisements
Advertisements
प्रश्न
State one relevant observation for the following:
At the anode, when molten lead bromide is electrolyzed using graphite electrodes.
उत्तर
Brown fumes of bromine gas are observed at the anode.
APPEARS IN
संबंधित प्रश्न
State the factors that influence the preferential discharge of ions at the electrodes.
Al+3 ,Cu+2 ,Na+ ,Zn+2 ions are present in aqueous solution, such that the concentration of ions is same, write the order of discharge of ions.
A metal article is to be electroplated with silver. The electrolyte selected is sodium argentocyanide.
- What kind of salt is sodium argentocyanide?
- Why is it preferred to silver nitrate as an electrolyte?
- State one condition to ensure that the deposit is smooth, firm and long lasting.
- Write the reaction taking place at the cathode.
- Write the reaction taking place at the anode.
Fill in the blank from the choices given below :
A molecule of _____ contains a triple bond.
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
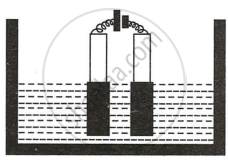
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
State one observation when electricity is passed through molten lead bromide.
Give appropriate scientific reasons for the following statement :
The electrical conductivity of acetic acid is less in comparision to the electrical conductivity of dilute sulphuric acid at a given concentration.
State the observation at the anode and at the cathode during the electrolysis of :
Fused lead bromide using graphite electrodes
Select the ion, that would get selectively discharge from the aqueous mixture of the ions listed below :
\[\ce{SO^{2-}_{4}}\], \[\ce{NO^{-}_{3}}\], \[\ce{OH-}\]
Classify the following substance:
Dilute sulphuric acid
