Advertisements
Advertisements
प्रश्न
State your observation when:
Decomposition of bicarbonates by dil. H2SO4
2NaHCO3 + H2SO4 → Na2SO4 + 2H2O + 2CO2
2KHCO3 + H2SO4 → K2SO4 + 2H2O + 2CO2
उत्तर
Dilute sulphuric acid liberates carbon dioxide from metallic carbonates and bicarbonates. Carbon dioxide, when bubbled into a test tube containing calcium hydroxide solution, turns it milky.
APPEARS IN
संबंधित प्रश्न
Potassium sulfite on reacting with hydrochloric acid releases _______ gas. (Cl2, SO2, H2S)
Give a chemical test to distinguish between the given pairs of chemicals:
Sodium chloride solution and Sodium nitrate solution
Write an equation for the reaction of hydrochloric acid on zinc carbonate.
Name an element which reacts with hydrogen to form a compound which is strongly acidic in water.
State which component is the oxidizing agent in aqua regia.
State the use of aqua-regia.
How will you distinguish between the two black samples, CuO and MnO2 with a chemical test?
Give a chemical test to distinguish between the following pair of compounds:
Sodium chloride solution and sodium nitrate solution.
A solution of hydrogen chloride in water is prepared. The following substances are added to separate portions of the solution :
| S.NO. | Substances added | Gas evolved | Odour |
| 1. | Calcium carbonate | ______ | ______ |
| 2. | Magnesium ribbon | ______ | ______ |
| 3. | Manganese (IV) oxide with heating | ______ | ______ |
| 4. | Sodium sulphide | ______ | ______ |
Complete the table by writing the gas evolved in each case and its odour.
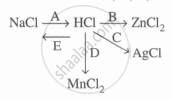
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.