Advertisements
Advertisements
प्रश्न
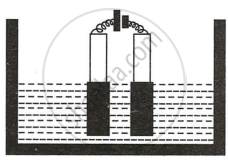
Study the diagram given alongside and answer the questions that follow :
(i) Give the names of the electrode A and B.
(ii) Which electrode is oxidizing electrode?
उत्तर
(i) Electrode A - Anode
Electrode B - Cathode
(ii) Electrode A.
APPEARS IN
संबंधित प्रश्न
Define the term : Electrolyte
Make a neatly labeled sketch to show how a brass spoon can be plated with silver. Explain your choice of electrolyte used.
Amongst the OH- ions and Br- ions which are likely to discharge first?
If a fused metallic chloride is electrolyzed, at which electrode would the metal be obtained?
Explain, why copper though a good conductor of electricity is, a non- electrolyte.
A strip of copper is placed in four different colourless salt solutions. They are KNO3, AgNO3, Zn(NO3)2, Ca(NO3)2. Which one of the solutions will finally turn blue?
Why is carbon tetrachloride, which is a liquid, a non-electrolyte?
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
Name the product formed at the anode during the electrolysis of acidified water using platinum electrodes
Copper sulphate solution is electrolysed using copper electrodes.
Write the equation for the reaction occurring at the Anode electrode.
