Advertisements
Advertisements
प्रश्न
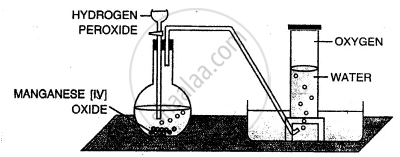
Taking hydrogen peroxide, how would you prepare oxygen in the laboratory ?
उत्तर
Take manganese dioxide in a round bottom flask and add hydrogen peroxide drop by drop to it, which acts ; a catalyst as shown in the figure. Collect oxygen by downward displacement of water.
APPEARS IN
संबंधित प्रश्न
Why is hydrogen peroxide preferred in the preparation of oxygen gas ?
Fill in the blank space by choosing the correct word from the given list.
List: oxygen, carbonic, suIphur dioxide gas, precipitation, helium
Every nine parts of water by weight contains eight parts by weight of _________.
What happens when Magnesium burns in oxygen ? Also give the balanced chemical equation for the reaction.
Give balanced equation to obtain oxygen from Silver oxide.
How will you identify oxygen in the laboratory ?
Complete and balance the following equation
S + O2 →
Tick (√ ) the most appropriate answer.
Sodium burns in oxygen with a flame which is :
Mention the physical properties of oxygen.
Oxygen is a poor conductor of heat and electricity.
Write a note on the rusting process.
