Advertisements
Advertisements
प्रश्न
The electronic configuration of metal A is 2, 8, 18, 1.
The metal A when exposed to air and moisture forms B a green layered compound. A with con. H2 SO4 forms C and D along with water. D is a gaseous compound. Find A, B, C, and D.
उत्तर
(i) The electronic configuration of metal (A) is 2, 8, 18, 1. A is copper (Z = 29)
(ii) (A) Copper exposed to air and moisture forms a green layered compound (B) that is copper carbonate.
\[\ce{\underset{\text{(A)}}{2Cu} + O2 + CO2 + H2O-> CuCO3.\underset{\text{(B)}}{Cu(OH)2}}\]
(iii) Copper (A) reacts with conc.H2SO4 to give copper sulphate (C) and Sulphur dioxide (D).
\[\ce{\underset{\text{(A)}}{Cu} + conc.2H2SO4->\underset{\text{(C)}}{CuSO4} + \underset{\text{(D)}}{SO2↑} + 2H2O}\]
| A | Copper | Cu |
| B | Copper carbonate | \[\ce{CuCO3.Cu(OH)2}\] |
| C | Copper sulphate | CuSO4 |
| D | Sulphur dioxide | SO2 |
APPEARS IN
संबंधित प्रश्न
Arrange the following as per the instruction given in the brackets:
Na, K, Li (Increasing atomic size)
Why is the size of sodium is greater than magnesium?
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
Among the elements of the second period, Li to Ne, pick out the element with the largest atomic size
Fill in the blanks.
Size of the atoms ______ from left to right across a period and ______ on descending in a group of normal elements.
Arrange the following in increasing order of property indicated
I, I+, I- (atomic size)
Write scientific reason.
In same period, boron and oxygen elements have different atomic size.
Which of the following is the correct order of atomic size?
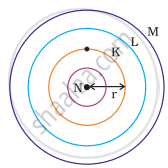
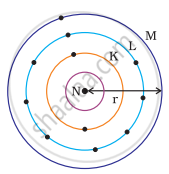
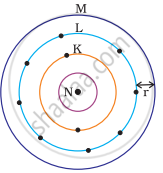
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Arrange the following as per the instruction given in the bracket:
Carbon, Fluorine, Beryllium (decreasing order of atomic size).