Advertisements
Advertisements
प्रश्न
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
उत्तर
Name: Helium
Symbol: He
Explanation:
Helium (He) is the noble gas with the smallest atomic radius. Although noble gases have complete outer electron shells and do not readily form bonds, helium has the smallest atomic size because it has only two electrons and experiences strong nuclear attraction, pulling the electrons closer to the nucleus.
संबंधित प्रश्न
Answer the following in respect of element `31/15 P `
Is it a reducing agent or oxidizing agent?
Why is the size of neon greater than fluorine?
Which is greater in size an atom or a cation?
Which is greater in size Fe2+ or Fe3+?
Give reasons for the following:
The size of the anion is greater than the size of the parent atom.
With reference to the variation of properties in the Periodic Table, which of the following is generally true?
Atomic size increases from left to right across a period.
The following questions refer to the Periodic Table.
What happens to the atomic size of elements moving from top to bottom of a group?
Arrange the following as per the instruction given in the bracket.
Mg, Cl, Na, S, Si (increasing order of atomic size)
Fill in the blank by selecting the correct word from the bracket.
If an element has seven electrons in its outermost shell then it is likely to have the _____ atomic size among all the elements in the same period.
The changes in the properties of elements on moving from left to right across a period of the Periodic Table. For the property, choose the correct answer.
The atomic size:
_______ is the distance between the nucleus of the atom and its outermost shell.
The electronic configuration of metal A is 2, 8, 18, 1.
The metal A when exposed to air and moisture forms B a green layered compound. A with con. H2 SO4 forms C and D along with water. D is a gaseous compound. Find A, B, C, and D.
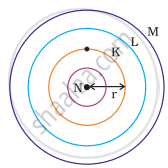
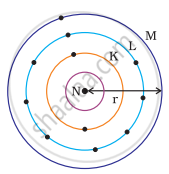
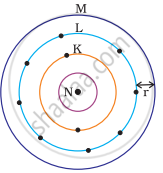
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Atomic number of a few elements are given below
10, 20, 7, 14
- Identify the elements
- Identify the Group number of these elements in the Periodic Table
- Identify the Periods of these elements in the Periodic Table
- What would be the electronic configuration for each of these elements?
- Determine the valency of these elements
- Electropositive nature of the element(s) increases down the group and decreases across the period
- Electronegativity of the element decreases down the group and increases across the period
- Atomic size increases down the group and decreases across a period (left to right)
- Metallic character increases down the group and decreases across a period.
On the basis of the above trends of the Periodic Table, answer the following about the elements with atomic numbers 3 to 9.
- Name the most electropositive element among them
- Name the most electronegative element
- Name the element with smallest atomic size
- Name the element which is a metalloid
- Name the element which shows maximum valency.
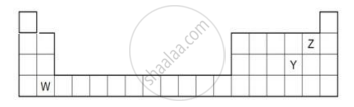
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
An element with the largest atomic radius among the following is ______.
In Period 3 of the Periodic Table, element B is placed to the left of element A. On the basis of this information, choose the correct word from the option to complete the following statement:
The element A would have ______ atomic size than B.