Advertisements
Advertisements
प्रश्न
उत्तर
Within a group the element with the largest size is expected to be present at the bottom of the group.
APPEARS IN
संबंधित प्रश्न
Give the trends in atomic size on moving across the period left to right.
Give reasons for the following:
The size of the anion is greater than the size of the parent atom.
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
Fill in the blank by selecting the correct word from the bracket.
If an element has seven electrons in its outermost shell then it is likely to have the _____ atomic size among all the elements in the same period.
An element X has mass number 40 and contains 21 neutrons in its atom. To which group of the Periodic Table does it belong?
Which of the following is the correct order of size?
When an atom of iodine becomes an iodine ion (I–) the radius will ______
Which of the following are the characteristics of isotopes of an element?
- Isotopes of an element have same atomic masses
- Isotopes of an element have same atomic number
- Isotopes of an element show same physical properties
- Isotopes of an element show same chemical properties
Atomic number of a few elements are given below
10, 20, 7, 14
- Identify the elements
- Identify the Group number of these elements in the Periodic Table
- Identify the Periods of these elements in the Periodic Table
- What would be the electronic configuration for each of these elements?
- Determine the valency of these elements
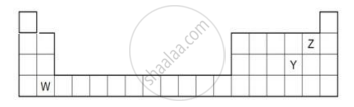
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?