Advertisements
Advertisements
प्रश्न
Within a group, where would you expect to find the element with the greatest metallic character.
उत्तर
Within a group the element with the greatest metallic character is expected to be present at the bottom of the group.
APPEARS IN
संबंधित प्रश्न
In Period 3 of the Periodic Table, element B is placed to the left of element A. On the basis of this information, choose the correct word from the option to complete the following statement:
The element B would have ______ metallic character than A.
Answer the following in respect of element `31/15 P `
What is its valency?
Give the trend in metallic character:
down the group top to bottom.
How does the chemical reactivity of
alkali metals vary?
iii) Observe the figure and answer the following questions.
a) Identify the block shown by box A and write an electronic configuration of any one element of this block.
b) Identify the block of element denoted by letter B and write its period number.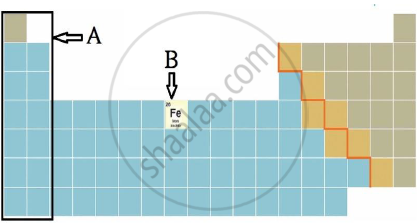
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 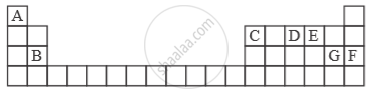
Which non-metallic element has the valency of 2?
Select the correct answer
Configuration of an element is 2, 8, 1. Which of the following statement is true?
Write scientific reason.
The metallic character of elements increases while going down the groups.
Which of the following set of elements is written in order of their increasing metallic character?
Choose the odd one out and write the reason:
