Advertisements
Advertisements
प्रश्न
Write scientific reason.
The metallic character of elements increases while going down the groups.
उत्तर
- While going down a group a new shell gets added, resulting in an increase in the distance between the nucleus and the valence electrons.
- This results in lowering the effective nuclear charge and thereby lowering the attractive force on the valence electrons.
- As a result of this, the tendency of the atom to form cation by losing electrons increases.
- The metallic character of an atom is its tendency to lose electrons.
Hence, the metallic character of elements increases while going down the groups.
APPEARS IN
संबंधित प्रश्न
Answer the following in respect of element `31/15 P `
Is it a metal or non - metal
An element barium has atomic number 56. Look up its position in the periodic table and answer the following question.
Is it a metal or a non-metal?
Arrange the elements of group 17 and group 1 according to the given conditions.
Increasing non – metallic character
A metal M forms as oxide having the formula M2O3. It belongs to third period. Write the atomic number and valency of the metal.
Choose the correct answer.
An alkaline earth metal
(i) Lead
(ii) potassium
(iii) calcium
(iv) Copper
Write scientific reason.
Metallic character goes on decreasing while going from left to right in a period.
In Period 3, the most metallic element is ______.
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 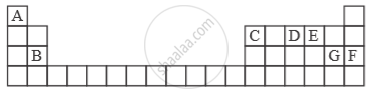
Which non-metallic element has the valency of 2?
Among the elements of the second period, Li to Ne, pick out the element that is the most reactive metal
For the main group of the periodic table, the metallic properties of the elements vary approximately with their position as shown in the table.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| H | He | ||||||
| A | B | ||||||
| C | D |
Will the most non-metallic element be found at A,B,C or D ?
State whether the following statement is true or false
All members of zero group are non metals.
Select the correct answer
Configuration of an element is 2, 8, 1. Which of the following statement is true?
Refering to changes in the properties of elements on moving from left to right across a period of the periodic table. Choose the correct answer.
The non-metallic character of the elements :
Arrange the following elements in order of their decreasing metallic character.
Na, Si, Cl, Mg, Al
The tendency of an element to form cation is the _______ character of that element.
Electropositivity means _______.
Write the name.
Two elements having two orbits.
Silicon is a metallic element.
3, 1, 2 electrons are in valence shells of X, Y, Z elements. From this information, state the group in which they belong and write their valencies.
Identify the elements with the following property and arrange them in increasing order of their reactivity
- An element which is a soft and reactive metal
- The metal which is an important constituent of limestone
- The metal which exists in liquid state at room temperature
