Advertisements
Advertisements
प्रश्न
Write scientific reason.
The non-metallic character increases while going from left to right in a period.
उत्तर
- While going from left to right in a period, electrons are added to the same outermost shell. At the same time, protons get added in the nucleus increasing the nuclear charge.
- Hence, these electrons experience greater pull from the nucleus due to increased effective nuclear charge.
- As a result, the ability of an atom to become anion by accepting outside electrons increases.
- The non-metallic character of an atom is its tendency to gain electrons.
Hence, non-metallic character increases while going from left to right in a period.
APPEARS IN
संबंधित प्रश्न
Arrange the following as per the instruction given in the bracket.
Cs, Na, Li, K, Rb (increasing order of metallic character).
Arrange the following as per the instructions given in the brackets:
Na, K, Cl, S, Si (increasing order ionization energy)
Give the trend in metallic character:
(i) across the period left to right
Give the trend in metallic character:
down the group top to bottom.
Name the periodic property which relates to the character of element which loses one or more electrons when supplied with energy.
Explain the following:
Group 17 elements are strong non-metals, while group 1 elements are strong metals.
How does the chemical reactivity of
alkali metals vary?
An element X belong to 3rd periods and group II of the periodic table state:
whether it is a metal or a non-metal.
The elements at the bottom of a group would be expected to show ______ metallic character than the element at the top.
Which has the maximum metallic character Na, Li or K?
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 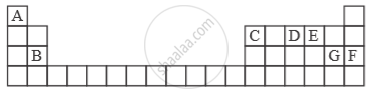
Which non-metallic element has the valency of 2?
For the main group of the periodic table, the metallic properties of the elements vary approximately with their position as shown in the table.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| H | He | ||||||
| A | B | ||||||
| C | D |
Will the most metallic element be found at A,B,C or D ?
Explain
Group 17 elements are non-metals, while group 1 elements are metals.
Select the correct answer
Configuration of an element is 2, 8, 1. Which of the following statement is true?
Copy and complete the following sentence choosing the correct word or words from those given below, at the end of the sentence:
The element at the bottom of a group would be expected to show ______ metallic character than the element at the top.
Electropositivity means _______.
Silicon is a metallic element.
3, 1, 2 electrons are in valence shells of X, Y, Z elements. From this information, state the group in which they belong and write their valencies.
An element has atomic number 17. To which group, the period does it belong? It is metal or non-metal?
