Advertisements
Advertisements
प्रश्न
Arrange the following as per the instruction given in the bracket.
Mg, Cl, Na, S, Si (increasing order of atomic size)
उत्तर
Mg, Cl, Na, S, Si (increasing order of atomic size) -
Cl < S < Si < Mg < Na
99 pm < 104 pm < 117 pm < 160 pm < 186 pm
APPEARS IN
संबंधित प्रश्न
Give the trends in atomic size on moving across the period left to right.
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
Write scientific reason.
Atomic radius goes on increasing down a group.
The electronic configuration of metal A is 2, 8, 18, 1.
The metal A when exposed to air and moisture forms B a green layered compound. A with con. H2 SO4 forms C and D along with water. D is a gaseous compound. Find A, B, C, and D.
Which of the following gives the correct increasing order of the atomic radii of O, F, and N?
Which of the following gives the correct increasing order of the atomic radii of O, F and N?
Which among the following elements has the largest atomic radii?
Atomic number of a few elements are given below
10, 20, 7, 14
- Identify the elements
- Identify the Group number of these elements in the Periodic Table
- Identify the Periods of these elements in the Periodic Table
- What would be the electronic configuration for each of these elements?
- Determine the valency of these elements
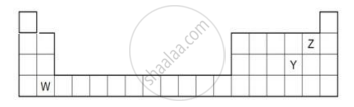
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
Arrange the following in order of increasing radii:
Cl−, Cl
Explain your choice.
