Advertisements
Advertisements
प्रश्न
The changes in the properties of elements on moving from left to right across a period of the Periodic Table. For the property, choose the correct answer.
The atomic size:
पर्याय
Decreases
Increases
Remains the same
Sometimes increases and sometimes decreases
उत्तर
Decreases
APPEARS IN
संबंधित प्रश्न
Which is greater in size an atom or a cation?
Write the name and symbol of the element from the description.
The atom having the smallest atomic mass.
Arrange the following as per the instruction given in the bracket
Na, K, Li (Increasing atomic size)
Atomic radius is expressed in the unit _______.
Write scientific reason.
In same period, boron and oxygen elements have different atomic size.
The electronic configuration of metal A is 2, 8, 18, 1.
The metal A when exposed to air and moisture forms B a green layered compound. A with con. H2 SO4 forms C and D along with water. D is a gaseous compound. Find A, B, C, and D.
Carbon belongs to the second period and Group 14. Silicon belongs to the third period and Group 14. If the atomic number of carbon is 6, the atomic number of silicon is ______
Which of the following gives the correct increasing order of the atomic radii of O, F and N?
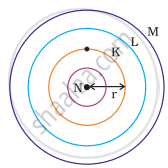
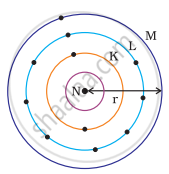
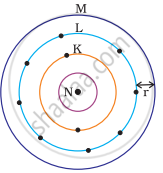
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Arrange the following elements in increasing order of their atomic radii.
- Li, Be, F, N
- Cl, At, Br I