Advertisements
Advertisements
प्रश्न
Arrange the following as per the instruction given in the bracket
Na, K, Li (Increasing atomic size)
उत्तर
Li < Na < K
APPEARS IN
संबंधित प्रश्न
Answer the following in respect of element `31/15 P `
Give its formula with chlorine?
What do you understand by atomic size? State its unit?
Which is greater in size Fe2+ or Fe3+?
Supply the missing word from those in the brackets:
If an element has seven electrons in its outermost shell then it is likely to have the ______ (largest/ smallest) atomic size among all the elements in the same period.
The metals of Group 2 from top to bottom are Be, Mg, Ca, Sr and Ba. Which one of these elements will form ions most readily and why?
While going from top to bottom in a group the atomic radius _______.
The electronic configuration of metal A is 2, 8, 18, 1.
The metal A when exposed to air and moisture forms B a green layered compound. A with con. H2 SO4 forms C and D along with water. D is a gaseous compound. Find A, B, C, and D.
Which of the following is the correct order of atomic size?
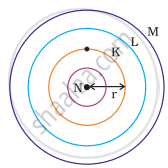
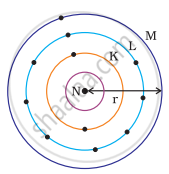
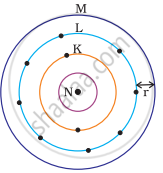
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Arrange the following as per instruction given in the bracket.
Mg, Cl, Na, S, Si (decreasing order of atomic size)