Advertisements
Advertisements
प्रश्न
The electrons in the atoms of four elements A, B, C and D are distributed in three shells having 1, 3, 5 and 7 electrons in the outermost shell respectively. State the period in which these elements can be placed in the modern periodic table. Write the electronic configuration of the atoms of A and D and the molecular formula of the compound formed when A and D combine.
उत्तर
| Elements | Valence electrons | Period |
| A | 1 | 3 |
| B | 3 | 3 |
| C | 5 | 3 |
| D | 7 | 3 |
Electronic configuration of A: 1s2 2s2 2p6 3s1
Electronic configuration of D: 1s2 2s2 2p6 3s2 3p5
Molecular formula of the compound formed with A and D:
Atomic number of A = 11
Electronic configuration A = 2,8,1
Number of valence electrons of A = 1
Valency of A = 1
Atomic number of D = 17
Electronic configuration D = 2,8,7
Number of valence electrons of D = 7
Valency of D = 8-7 = 1
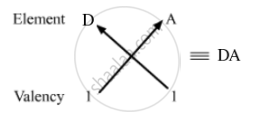
So, the formula of the compound formed when elements A and D combine is AD.
APPEARS IN
संबंधित प्रश्न
What happens to the metallic character of the elements as we go down in a group of the periodic table?
Which of the following element would lose an electron easily?
(a) Mg
(b) Na
(c) K
(d) Ca
Where would you locate the element with electronic configuration 2, 8 in the modern periodic table?
Correct the statements.
Elements in the same periods have equal valency.
Fill in the blank
Oxidising property ________from fluroine to iodine because the power to accpet electron decreases.
The following questions refer to the Periodic Table.
Name the first and last element in period 2.
The position of elements A, B, C, D and E in the periodic table are shown below:
|
Group 1 |
Group 2 |
Group 17 |
Group 18 |
|
|
|
|
D |
|
|
B |
C |
|
|
A |
|
|
E |
State which is most reactive (i) metal (ii) non-metal
Name the elements in Period 1.
What is meant by a Group in the Periodic Table?
Name the elements in the correct order of their increasing atomic numbers present in the first, second, and third short periods of the periodic table.
State which of the elements are –
- metallic
- non-metallic
- noble gases
in each of the periods 2 and 3.
