Advertisements
Advertisements
Question
The electrons in the atoms of four elements A, B, C and D are distributed in three shells having 1, 3, 5 and 7 electrons in the outermost shell respectively. State the period in which these elements can be placed in the modern periodic table. Write the electronic configuration of the atoms of A and D and the molecular formula of the compound formed when A and D combine.
Solution
| Elements | Valence electrons | Period |
| A | 1 | 3 |
| B | 3 | 3 |
| C | 5 | 3 |
| D | 7 | 3 |
Electronic configuration of A: 1s2 2s2 2p6 3s1
Electronic configuration of D: 1s2 2s2 2p6 3s2 3p5
Molecular formula of the compound formed with A and D:
Atomic number of A = 11
Electronic configuration A = 2,8,1
Number of valence electrons of A = 1
Valency of A = 1
Atomic number of D = 17
Electronic configuration D = 2,8,7
Number of valence electrons of D = 7
Valency of D = 8-7 = 1
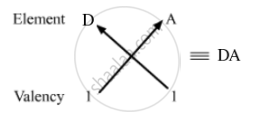
So, the formula of the compound formed when elements A and D combine is AD.
APPEARS IN
RELATED QUESTIONS
An element 'M' with electronic configuration (2, 8, 2) combines separately with (NO3)–, (SO4)2– and (PO4)3– radicals. Write the formula of the three compounds so formed. To which group and period of the Modern Periodic Table does the element 'M' belong? Will 'M' form covalent or ionic compounds? Give reason to justify your answer.
Two elements ‘A’ and ‘B’ belong to the 3rd period of Modern periodic table and are in group 2 and 13, respectively. Compare their following characteristics in tabular form:-
(a) Number of electrons in their atoms
(b) Size of their atoms
(c) Their tendencies to lose electrons
(d) The formula of their oxides
(e) Their metallic character
(f) The formula of their chlorides
Write the atomic numbers of two elements 'X' and 'Y' having electronic configuration 2, 8, 2 and 2, 8, 6 respectively.
What is meant by 'group' in the modern periodic table? How do the following change on moving from top to bottom in a group?
(i) Number of valence electrons
(ii) Number of occupied shells
(iii) Size of atoms
(iv) Metallic character of element
(v) Effective nuclear charge experienced by valence electrons
Calculate the number of gram atoms in 4.6 grams of sodium (Na = 23)
K, Pb, Ca, Zn (In the increasing order of the reactivity)
What is modern periodic law?
The elements of one short period of the periodic table are given below in order from left to right:
| Li | Be | B | C | O | F | Ne |
Place the three elements fluorine, beryllium and nitrogen in the order of increasing electronegativity.
With reference to the variation of properties in the Periodic table, which of the following is generally true?
Select the correct answer from the options given below.
It is a period having elements from atomic no. 11 to 18.
