Advertisements
Advertisements
प्रश्न
The equation for the reaction when compound A is bubbled through bromine dissolved in carbon tetrachloride is as follows:
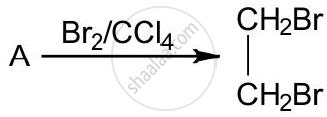
1) Draw the structure of A.
2) State your observation during this reaction.
उत्तर
1) CH2 = CH2
2) When bromine dissolved in CCl4 is added to ethene, the orange color of bromine disappears because of the formation of colorless ethylene bromide.
APPEARS IN
संबंधित प्रश्न
Name a few sources of organic chemistry ?
Fill in the blank:
More than 90% of the known organic compounds are. ________
Give the reactions of ethyne with Bromine.
LPG contains
Fill in the blanks by chosing words from the given list of words:
Addition, Carbohydrates, CnH2n-2, CnH2n+2, electrochemical, homologus, hydrocarbons, saturated, substitution, unsaturated.
The alkanes form a ______series with general formula_________The alkanes are_______,which generally undergo________reactions.
Compound A is bubbled through bromine dissolved in carbon tetrachloride and the product is CH2Br - CH2Br.
\[\ce{A ->[Br2/Od4] CH2Br - CH2Br}\]
What the procedure for converting the product of (b) (IV) back to A?
Give reason for the following :
Methane does not undergo addition reaction, but ethane does
Give balanced equation for the laboratory preparations of the following organic compound:
A saturated hydrocarbon from iodomethene
Give the structural formulae of the following:
2-methyl propane
Give the IUPAC name of the organic compound represented by the structural formula given below:
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{Cl}\phantom{..}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{Cl}\phantom{..}\ce{H}\phantom{...}\ce{H}\end{array}\]
