Advertisements
Advertisements
प्रश्न
The equation for the reaction when compound A is bubbled through bromine dissolved in carbon tetrachloride is as follows:
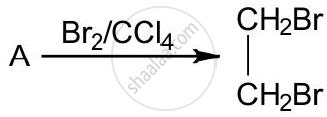
1) Draw the structure of A.
2) State your observation during this reaction.
उत्तर
1) CH2 = CH2
2) When bromine dissolved in CCl4 is added to ethene, the orange color of bromine disappears because of the formation of colorless ethylene bromide.
APPEARS IN
संबंधित प्रश्न
Which was the first organic compound synthesized in laboratory and by whom?
What are the main sources of alkanes and why they are known as saturated hydrocarbons?
Write the equations to show what happens when ethyne gas reacts with
Bromine water
Give the reactions of ethyne with Water.
Methane reacts with energy of chlorine in diffused sunlight to give the final product as
Name the addition product formed between ethane and water.
Name the organic compound prepared by each of the following reactions:
\[\ce{CaC2 + 2H2O ->}\]
Compound X bubbled through bromine dissolved in carbon tetrachloride CCl4;
\[\begin{array}{cc}
\ce{X ->[Br2CCl4] CH2Br}\\
\phantom{......}|\\
\phantom{...........}\ce{CH2Br}\\
\end{array}\]
Draw the structure of X.
Give the IUPAC name of the organic compound represented by the structural formula given below:
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{Cl}\phantom{..}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{Cl}\phantom{..}\ce{H}\phantom{...}\ce{H}\end{array}\]
