(English Medium)
Academic Year: 2015-2016
Date & Time: 18th March 2016, 11:00 am
Duration: 2h
Advertisements
Question 1 is Compulsory.
Attempt any four from Question 2 to Question 7
Choose the word or phrase from the brackets which correctly complete the following statement:
Metals are good ______ (oxidizing agents/reducing agents) because they are electron ______ (acceptors/donors).
Chapter: [0.01] Periodic Table, Periodic Properties and Variations of Properties
Electrovalent compounds have ________________ (high / low) melting points.
Chapter:
Higher the pH value of a solution, the more ______ it is.
Acidic
Alkaline
Chapter: [0.03] Study of Acids, Bases and Salts [0.1] Practical Work
_____________ (AgCl / PbCl2), a white precipitate is soluble in excess NH4OH
Chapter: [0.04] Analytical Chemistry
Conversion of ethene to ethane is an example of ______.
Hydration
Hydrogenation
Chapter: [0.09] Organic Chemistry
An element with the atomic number 19 will most likely combine chemically with the element whose atomic number is ______.
17
11
28
20
Chapter: [0.01] Periodic Table, Periodic Properties and Variations of Properties
Choose the correct answer from the options given below:
The ratio between the number of molecules in 2g of hydrogen and 32g of oxygen is:
[Given that H = 1, O = 16]
1: 2
1: 0.01
1: 1
0.01: 1
Chapter: [0.05] Mole Concept and Stoichiometry
Choose the correct answer from the options given below :
The two main metals in Bronze are:
(A) Copper and zinc
(B) Copper and lead
(C) Copper and nickel
(D) Copper and tin
Chapter: [0.07] Metallurgy
Choose the correct answer from the options given below :
The particles present in strong electrolytes are :
(A) Only molecules
(B) Mainly ions
(C) Ions and molecules
(D) Only atoms
Chapter: [0.06] Electrolysis
The aim of the Fountain experiment is to prove that ______.
\[\ce{HCl}\] turns blue litmus red.
\[\ce{HCl}\] is denser than air.
\[\ce{HCl}\] is highly soluble in water.
\[\ce{HCl}\] fumes in moist air.
Chapter: [0.081] Hydrogen Chloride
Write balanced chemical equations for Action of warm water on AIN.
Chapter: [0.08199999999999999] Ammonia [0.1] Practical Work
Write balanced chemical equations for Action of hot and concentrated Nitric acid on copper
Chapter: [0.083] Nitric Acid
Write a balanced chemical equation for the action of hydrochloric acid on sodium bicarbonate.
Chapter: [0.081] Hydrogen Chloride [0.1] Practical Work
Write balanced chemical equations for the action of dilute sulphuric acid on sodium sulphite.
Chapter: [0.084] Sulphuric Acid
Write balanced chemical equations for Preparation of ethanol from Ethyl Chloride
Chapter: [0.09] Organic Chemistry
State your observation when dilute hydrochloric acid is added to a lead nitrate solution and the mixture is heated.
Chapter: [0.081] Hydrogen Chloride
State your observations when Barium chloride solution is mixed with Sodium Sulphate Solution.
Chapter: [0.04] Analytical Chemistry
State your observations when Concentrated Sulphuric acid is added to Sugar Crystals
Chapter: [0.084] Sulphuric Acid
State your observations when Dilute Hydrochloric acid is added to Copper carbonate
Chapter: [0.081] Hydrogen Chloride
State your observations when Dilute Hydrochloric acid is added to Sodium thiosulphate.
Chapter: [0.081] Hydrogen Chloride
Identify the term/substance in the following:
The tendency of an atom to attract electrons to itself when combined in a compound.
Chapter: [0.01] Periodic Table, Periodic Properties and Variations of Properties
Identify the term/substance in the following:
The method used to separate ore from gangue by preferential wetting
Chapter: [0.07] Metallurgy
Identify the term/substance in the following:
The catalyst used in the conversion of ethyne to ethane.
Chapter: [0.09] Organic Chemistry
Identify the term/substance in the following:
The type of reactions alkenes undergo.
Chapter: [0.09] Organic Chemistry
Identify the term/substance in the following:
The electrons present in the outermost shell of an atom.
Chapter: [0.02] Chemical Bonding
A gas of mass 32 gms has a volume of 20 liters at S.T.P. Calculate the gram molecular weight of the gas.
Chapter: [0.05] Mole Concept and Stoichiometry
How much Is Calcium oxide formed when 82g of calcium nitrate is heated? Also, find the volume of nitrogen dioxide evolved :
2Ca(NO3)2 → 2CaO+4NO2+O2
(Ca = 40 , N = 14,O = 16)
Chapter: [0.05] Mole Concept and Stoichiometry
Match the salts given in Column I with their method of preparation given in Column II
| Column I | Column II | ||
| 1 | Pb(NO3)2 from PbO | A | Simple displacement |
| 2 | MgCl2 from Mg | B | Titration |
| 3 | FeCl3 from Fe | C | Neutralization |
| 4 | NaNO3 from NaOH | D | Precipitation |
| 5 | ZnCO3 from ZnSO4 | E | Combination |
Chapter: [0.04] Analytical Chemistry
Rewrite the following sentences by using the correct symbol > (greater than) or < (less than) in the blanks given
The ionization potential of potassium is _________________ that of sodium.
Chapter: [0.01] Periodic Table, Periodic Properties and Variations of Properties
Rewrite the following sentences by using the correct symbol > (greater than) or < (less than) in the blanks given
The electronegativity of iodine is ___________ that of Chlorine
Chapter: [0.01] Periodic Table, Periodic Properties and Variations of Properties
Use the letters only written in the Periodic Table given below to answer the questions that Follow :

1) State the number of valence electrons in atom J.
2) Which element shown forms ions with a single negative charge?
3)Which metallic element is more reactive than R?
4) Which element has its electrons arranged in four shells?
Chapter: [0.01] Periodic Table, Periodic Properties and Variations of Properties
If an element has a low ionization energy then it is likely to be ______________ (metallic / non metallic).
Chapter: [0.07] Metallurgy
If an element has seven electrons in its outermost shell then it is likely to have the ______________ atomic size among all the elements in the same period.
Largest
Smallest
Chapter: [0.01] Periodic Table, Periodic Properties and Variations of Properties
Advertisements
The following table shows the electronic configuration of the elements W, X, Y, Z:
| Element | w | x | y | z |
| Electronic configurations |
2, 8, 1 | 2, 8, 7 | 2, 5 | 1 |
Answer the following questions based on the table above:
1) What type of Bond is formed between :
a) W and X
b) Y and Z
2) What is the formula of the compound formed between :
a) X and Z
b) W and X
Chapter: [0.01] Periodic Table, Periodic Properties and Variations of Properties
Write a balanced chemical equation for Burning of ethane in the plentiful supply of air.
Chapter: [0.09] Organic Chemistry
Write a balanced chemical equations for Action of water on Calcium carbide
Chapter:
Write a balanced chemical equations for Heating of Ethanol at 170°C in the presence of conc. Sulphuric acid
Chapter: [0.09] Organic Chemistry
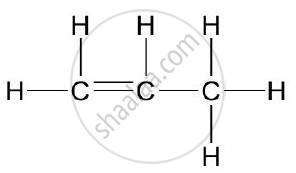
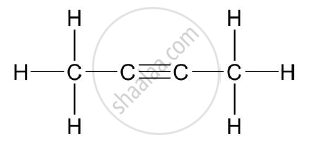
Give the structural formulae of 2-methylpropane.
Chapter: [0.09] Organic Chemistry
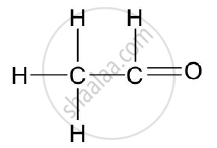
Give the structural formulae of Ethanoic acid.
Chapter: [0.09] Organic Chemistry
Give the structural formulae of Butan – 2 -ol
Chapter: [0.09] Organic Chemistry
The equation for the reaction when compound A is bubbled through bromine dissolved in carbon tetrachloride is as follows:
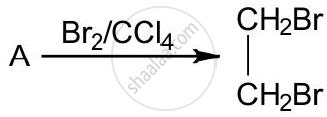
1) Draw the structure of A.
2) State your observation during this reaction.
Chapter: [0.09] Organic Chemistry
Fill in the blanks using the appropriate words given below :
(Sulphur dioxide, nitrogen dioxide, Nitric oxide, Sulphuric acid)
Cold, dilute nitric acid reacts with copper to give ___________.
Chapter: [0.083] Nitric Acid
Fill in the blanks using the appropriate words given below :
(Sulphur dioxide, nitrogen dioxide, Nitric oxide, Sulphuric acid)
Hot, concentrated nitric acid reacts with sulphur to form ______________.
Chapter: [0.083] Nitric Acid
Identify the gas evolved and give the chemical test in the following cases
Dilute hydrochloric acid reacts with sodium sulphite.
Chapter: [0.081] Hydrogen Chloride [0.1] Practical Work
Identify the gas evolved and give the chemical test in the following cases
Dilute hydrochloric acid reacts with iron (II) sulphide.
Chapter: [0.081] Hydrogen Chloride [0.1] Practical Work
State your observations when ammonium hydroxide solution is added drop by drop and then in excess to each of the following solutions :
1) Copper sulphate solution
2) Zinc sulfate solution.
Chapter: [0.04] Analytical Chemistry
Write equations for the reactions taking place at the two electrodes (mentioning clearly the name of the electrode) during the electrolysis of Acidified copper sulphate solution with copper electrodes
Chapter: [0.06] Electrolysis
Write equations for the reactions taking place at the two electrodes (mentioning clearly the name of the electrode) during the electrolysis of Molten lead bromide with inert electrodes
Chapter: [0.06] Electrolysis
Name the product formed at the anode during the electrolysis of acidified water using platinum electrodes.
Chapter: [0.06] Electrolysis
Name the metallic ions that should be present in the electrolyte when an article made of copper is to be electroplated with silver.
Chapter: [0.06] Electrolysis
Advertisements
A gas cylinder contains 12 x 1024 molecules of oxygen gas.
If Avogadro’s number is 6 x 1023; Calculate:
1)The mass of oxygen present in the cylinder.
2) The volume of oxygen at S.T.P. present in the cylinder. [O = 16]
Chapter: [0.05] Mole Concept and Stoichiometry
A gaseous hydrocarbon contains 82.76% of carbon. Given that its vapor density is 29, find its molecular formula. [C = 12, H = 11]
Chapter: [0.05] Mole Concept and Stoichiometry
The equation 4NH3 + 5O2 → 4NO + 6 H2O, represents the catalytic oxidation of ammonia. If 100 cm3 of ammonia is used calculate the volume of oxygen required to oxidize the ammonia completely
Chapter: [0.08199999999999999] Ammonia
By drawing an electron dot diagram show the formation of Ammonium ion [Atomic No.: N = 7 and H = 1]
Chapter: [0.02] Chemical Bonding
Name the gas evolved when the following mixtures are heated:
Calcium hydroxide and Ammonium Chloride
Chapter: [0.08199999999999999] Ammonia
Name the gas evolved when the following mixtures are heated:
Sodium Nitrite and Ammonium Chloride
Chapter: [0.083] Nitric Acid
Write balanced chemical equations for the following:
When the excess of ammonia is treated with chlorine.
Chapter: [0.08199999999999999] Ammonia
Write balanced chemical equations for the following:
An equation to illustrate the reducing nature of ammonia.
Chapter: [0.08199999999999999] Ammonia
A, B, C, and D summarize the properties of sulphuric acid depending on whether it is dilute or concentrated.
A = Typical acid property
B = Non volatile acid
C = Oxidizing agent
D = Dehydrating agent
Choose the property (A, B, C or D) depending on which is relevant to each of the following
1) Preparation of Hydrogen chloride gas
2) Preparation of Copper sulphate from copper oxide.
3) An action of conc. Sulphuric acid on Sulphur.
Chapter: [0.084] Sulphuric Acid
Give reasons why Sodium Chloride will conduct electricity only in the fused or aqueous solution state.
Chapter: [0.06] Electrolysis
Give reasons why In the electroplating of an article with silver, the electrolyte sodium argentocyanide solution is preferred over silver nitrate solution.
Chapter: [0.06] Electrolysis
Give reasons why Although copper is a good conductor of electricity, it is a non-electrolyte.
Chapter: [0.06] Electrolysis
Name the solution used to react with Bauxite as a first step in obtaining pure aluminum oxide, in the Baeyer’s process
Chapter: [0.07] Metallurgy
Write the equation for the reaction where the aluminum oxide for the electrolytic extraction of aluminum is obtained by heating aluminum hydroxide.
Chapter: [0.07] Metallurgy
Name the compound added to pure alumina to lower the fusion temperature during the electrolytic reduction of alumina.
Chapter: [0.07] Metallurgy
Write the equation for the reaction that occurs at the cathode during the extraction of aluminum by electrolysis.
Chapter: [0.07] Metallurgy
Explain why it is preferable to use a number of graphite electrodes as the anode instead of a single electrode, during the above electrolysis.
Chapter: [0.06] Electrolysis
State what would you observe when Washing Soda Crystals are exposed to the atmosphere
Chapter: [0.03] Study of Acids, Bases and Salts
State what would you observe when The salt ferric chloride is exposed to the atmosphere
Chapter: [0.03] Study of Acids, Bases and Salts
Identify the cations in the following case:
NaOH solution, when added to the Solution (A), gives a reddish brown precipitate
Chapter: [0.04] Analytical Chemistry
Identify the cations in the following case :
NH4OH Solution, when added to the Solution (B), gives white ppt which does not dissolve in excess.
Chapter: [0.08199999999999999] Ammonia
Identify the cations in the following case:
NaOH Solution, when added to Solution (C), gives white ppt which is insoluble in excess
Chapter: [0.08199999999999999] Ammonia
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CISCE previous year question papers ICSE Class 10 Chemistry with solutions 2015 - 2016
Previous year Question paper for CISCE ICSE Class 10 -2016 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CISCE ICSE Class 10 .
How CISCE ICSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.