Advertisements
Advertisements
प्रश्न
The SI unit of heat energy is ______.
पर्याय
joule
calorie
kilo calorie
Cal/g °C
none of these
Kcal/kg
उत्तर
The SI unit of heat energy is a joule.
Explanation:
The unit of heat in the SI system is the joule (J) and the calorie (cal) in the CGS measurement system. The heat required to raise the temperature of one kilogram of water by 1 °C from 14.5 °C to 15.5 °C is called one kilocalorie of heat.
APPEARS IN
संबंधित प्रश्न
Select the correct alternative
A thermometer uses
What is heat ? State its S.I. unit.
Define the term heat.
Define the term calorie. How is it related to joule (the S.I. unit of heat)?
Cooking utensils are made from metals due to their property of ______.
What will absorb heat?
Glass vessel
What will absorb heat?
Plastic plate
The unit of heat is
The amount of heat gained by a substance is equal to the product of its mass and latent heat.
Calorie is the unit of ______.
We reduce the heat by adding ______ while preparing fruit juice.
In the past, people used to rub two wooden pieces together to light fire.
A circular metal loop is heated at point O as shown in Figure 4.6.
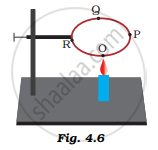
(i) In which direction would heat flow in the loop?
(ii) In which order are the pins at points P, Q and R fixed with the help of wax fall if points O, P, Q and R are equidistant from each other?
You would have noticed some space being left on railway tracks. Why?
Arrange the following in order of decreasing the expansion of heating: Steel, milk, air.
Which of the following is true?
The sum of the kinetic and potential energy is called the ______ of the molecules.
