Advertisements
Advertisements
प्रश्न
Under what conditions does ethane gets converted to Ethyl alcohol
उत्तर १
When a mixture of ethane and oxygen is compressed to about 120atm pressure and passed over copper tubes at 475K, ethyl alcohol is formed.
`2C_2H_6 +O_2` 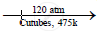 `2C_2H_5OH`
`2C_2H_5OH`
उत्तर २
When a mixture of ethane and oxygen is compressed to about 120atm pressure and passed over copper tubes at 475K, ethyl alcohol is formed.
\[\ce{2C2H6 + O2 ->[120atm][Cutubes,475K]2C2H5OH}\]
APPEARS IN
संबंधित प्रश्न
Identify the term/substance in the following:
The type of reactions alkenes undergo.
Write the structure of the following compound:
2 – methyl propane
Write the electron dot formula of ethane.
The molecules of the alkene family are represented by a general formula CnH2n. Answer the following:
What is the structural formula of the third member of the alkene family?
Give a balanced equation for the lab. Preparation of ethylene. How is the gas collected?
How is ethene prepared by a dehydration reaction? Give an equation and name the products formed.
Give the conditions and the main products formed by hydrogenation of ethene.
Give the formula and name of A, B, C and D in the following equation:
\[\ce{CH4 ->[Cl2][-HCl] A ->[Cl2][-HCl] B ->[Cl2][-HCl] C->[Cl2][-HCl] D}\]
Give the formula and name of A, B C and D in the following equations:
`C_2H_4 + Cl_2 → A `
Fill in blanks with correct words from the brackets:
Alkenes are the__________(Analogous/homologous) sries of _________(saturated/unsaturated) hydrocarbons. They differ from alkanes due to the presence of_______(double/single) bonds. Alkenes mainly undergo________(addition/substitution) reactions.
