Advertisements
Advertisements
Question
Under what conditions does ethane gets converted to Ethyl alcohol
Solution 1
When a mixture of ethane and oxygen is compressed to about 120atm pressure and passed over copper tubes at 475K, ethyl alcohol is formed.
`2C_2H_6 +O_2` 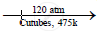 `2C_2H_5OH`
`2C_2H_5OH`
Solution 2
When a mixture of ethane and oxygen is compressed to about 120atm pressure and passed over copper tubes at 475K, ethyl alcohol is formed.
\[\ce{2C2H6 + O2 ->[120atm][Cutubes,475K]2C2H5OH}\]
APPEARS IN
RELATED QUESTIONS
Write the structure of the following compound:
2-methylprop-1-ene
Convert methyl iodide into ethane.
Write the electron dot formula of ethane.
The molecules of the alkene family are represented by a general formula CnH2n. Answer the following:
What is the molecular formula of alkene when n = 4?
How is ethene prepared by a dehydration reaction? Give an equation and name the products formed.
Write the following property of ethene:
Density as compared to air
How would you convert ethene into 1,2-dibromoethane?
Write the name and formula of the product formed in the case below:
\[\ce{H2C = CH2 + HBr}\] →
Write the equation for the following reaction:
Ethene and water (steam)
Give reasons:
Ethene is more reactive than ethane.
