Advertisements
Advertisements
प्रश्न
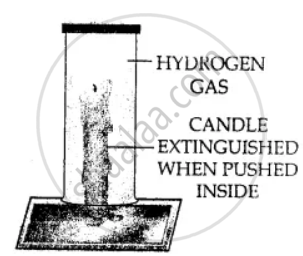
Using a burning candle and a jar of hydrogen – how would you prove experimentally that Hydrogen does not support combustion.
उत्तर
Hydrogen extinguishes the burning candle when pushed inside the jar. This shows that hydrogen does not support combustion.
APPEARS IN
संबंधित प्रश्न
Indicate which of the following statement is true and which is false:
Hydrogen is combustible as well as a supporter of combustion.
Give a balanced chemical equation for the reaction.
State four uses of hydrogen:
Write the balanced equation and give your observation when the following metal reacts:
Sodium with cold water
With reference to the equation : Cl2 + H2S → 2HCl + S pertaining to a redox reaction – select the correct answer.
Chlorine acts as an ______ agent.
Select the correct options for the following statement:
The gas which has now replaced hydrogen in air balloons is ___.
What do the following symbols [or formula] denote : 2H ; H2 ; H+. [two atoms, molecule, ion]
Explain the following:
Two jars of H2 are collected – “one burns quietly and the other does not”.
Write correctly the balanced equation for the following:
‘When zinc filings are added to a concentrated solution of sodium hydroxide’.
How would you obtain ‘hydrogen from sodium hydroxide’ solution other than by electrolysis?
