Advertisements
Advertisements
Question
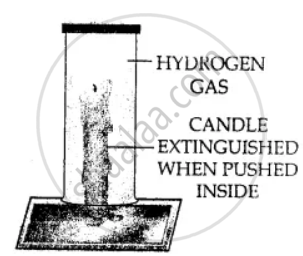
Using a burning candle and a jar of hydrogen – how would you prove experimentally that Hydrogen does not support combustion.
Solution
Hydrogen extinguishes the burning candle when pushed inside the jar. This shows that hydrogen does not support combustion.
APPEARS IN
RELATED QUESTIONS
Indicate which of the following statement is true and which is false:
Hydrogen is combustible as well as a supporter of combustion.
Indicate which of the following statement is true and which is false:
Hydrogen gas is easily liquefiable.
FILL IN THE BLANK
In nature, hydrogen occurs as a .................. molecule represented as H2
Why is hydrogen called a reducing agent? Give chemical equation to support your answer ?
Choose terms from the options given in brackets to complete this sentence.
A metal …………….. hydrogen in the activity series gives hydrogen with …………… acid or …………… acid.
(above, below, dilute hydrochloric, concentrated hydrochloric, dilute sulphuric).
Correct the following statement:
Hydrogen is dried by passing it through conc. H2SO4.
Give reason for the following:
The reaction of chlorine with hydrogen sulphide is deemed a redox reaction.
How can you obtain hydrogen from sodium hydroxide [not by electrolysis].
Write down the “word equation” for the following reaction:
sodium hydroxide solution + zinc → ?
Write balanced equation of the reaction in the preparation of : hydrogen from a solution of potassium hydroxide [other than by electrolysis].
