Advertisements
Advertisements
प्रश्न
What are the different types of thermodynamic systems?
उत्तर
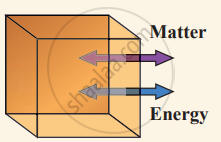
(i) Open system can exchange both matter and energy with the environment.
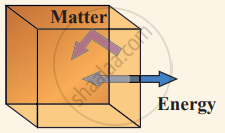
(ii) Closed system exchange energy but not matter with the environment.
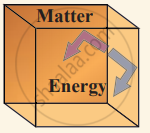
(iii) Isolated system can exchange neither energy nor matter with the environment.
APPEARS IN
संबंधित प्रश्न
A gas in a closed container is heated with 10 J of energy, causing the lid of the container to rise 2m with 3N of force. What is the total change in energy of the system?
On what basis a thermodynamic system can be classified?
An ideal gas passes from one equilibrium state (P1, V1, T1, N) to another equilibrium state (2P1, 3V1, T2, N). Then
A hot cup of coffee is kept on the table. After some time it attains a thermal equilibrium with the surroundings. By considering the air molecules in the room as a thermodynamic system, which of the following is true
What is meant by ‘thermal equilibrium’?
What is mean by state variable? Give example.
Discuss the thermal equilibrium.
Discuss the mechanical equilibrium.
Discuss the chemical equilibrium.
What are surroundings in thermodynamics?
