Advertisements
Advertisements
प्रश्न
What is the covalence of nitrogen in N2O5?
उत्तर
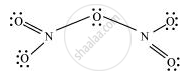
From the structure of N2O5, it is evident that the covalence of nitrogen is 4.
APPEARS IN
संबंधित प्रश्न
Write balanced chemical equations for action of potassium permanganate on: Hydrogen
Pb(NO3)2 on heating gives a brown gas which undergoes dimerisation on cooling? Identify the gas
Why does NO2 dimerise?
Bond angle in `PH_(4)^(+)` is higher than that in PH3. Why?
Why does the reactivity of nitrogen differ from phosphorus?
How is nitrogen prepared in the laboratory? Write the chemical equations of the reactions involved.
Explain why NH3 is basic while BiH3 is only feebly basic.
Which of the following is a basic oxide?
(A) SiO2
(B) P4O10
(C) MgO
(D) Al2O3
Write balanced chemical equations for action of potassium permanganate on Warm conc. sulphuric acid
The statement true for \[\ce{N^-_3}\] is ____________.
Which of the following statements is not correct for nitrogen?
Pick out the wrong statement.
Which of the following oxides of nitrogen reacts with FeSO4 to form a dark brown compound?
Which of the following elements can be involved in pπ – dπ bonding?
Name three oxoacids of nitrogen. Write the disproportionation reaction of that oxoacid of nitrogen in which nitrogen is in +3 oxidation state.
HCl is added to following oxides. Which one would give H2O2
Heating white phosphorus with conc. NaOH solution gives mainly ______.
Assertion: Nitrogen and oxygen are the main components in. the atmosphere but these do not react to form oxides of nitrogen.
Reason: The reaction between nitrogen and oxygen requires high temperature.
