Advertisements
Advertisements
Question
What is the covalence of nitrogen in N2O5?
Solution
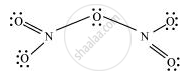
From the structure of N2O5, it is evident that the covalence of nitrogen is 4.
APPEARS IN
RELATED QUESTIONS
Write balanced chemical equations for action of potassium permanganate on: Hydrogen
Pb(NO3)2 on heating gives a brown gas which undergoes dimerisation on cooling? Identify the gas
Which of the following is a basic oxide?
(A) SiO2
(B) P4O10
(C) MgO
(D) Al2O3
Write balanced chemical equations for action of potassium permanganate on Warm conc. sulphuric acid
Tendency to show –3 oxidation state decrease from Nitrogn (N) to Bismuth (Bi).
The statement true for \[\ce{N^-_3}\] is ____________.
Among the 15th group elements, as we move from nitrogen to bismuth, the pentavalency becomes less pronounced and trivalency becomes more pronounced due to:
Pure nitrogen is prepared in the laboratory by heating a mixture of:
Which of the following oxides is neutral?
In which one of the following oxides of nitrogen, one nitrogen atom is not directly linked to oxygen?
Which oxide of nitrogen is obtained on heating ammonium nitrate at 250°C?
A deep brown gas is formed by mixing two colourless gases which are:
The laughing gas is ____________.
On heating lead nitrate forms oxides of nitrogen and lead. The oxides formed are ______.
HCl is added to following oxides. Which one would give H2O2
Which oxide of nitrogen is obtained on heating ammonium nitrate at 250°C?
