Advertisements
Advertisements
प्रश्न
What do you mean by internal resistance of a cell?
उत्तर
The resistance offered by the electrolyte of a cell to the flow of current between its electrodes is called internal resistance of the cell. An ideal battery has zero internal resistance and the potential difference across the battery equals to its emf. But a real battery is made of electrodes and electrolyte, there is resistance to the flow of charges within the battery. A freshly prepared cell has low internal resistance and it increases with ageing.
APPEARS IN
संबंधित प्रश्न
The internal resistance of a 2.1 V cell which gives a current of 0.2 A through a resistance of 10Ω is ______.
Explain the determination of the internal resistance of a cell using voltmeter.
Three identical lamps each having a resistance R are connected to the battery of emf as shown in the figure.
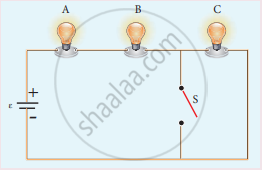
Suddenly the switch S is closed.
- Calculate the current in the circuit when S is open and closed.
- What happens to the intensities of the bulbs A, B and C.
- Calculate the voltage across the three bulbs when S is open and closed.
- Calculate the power delivered to the circuit when S is opened and closed.
- Does the power delivered to the circuit decrease, increase or remain same?
A cell supplies a current of 0.9 A through a 2 Ω resistor and a current of 0.3 A through a 7 Ω resistor. Calculate the internal resistance of the cell.
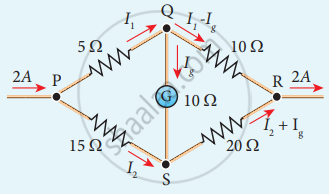
Determine the current flowing through the galvanometer (G) as shown in the figure.