Advertisements
Advertisements
प्रश्न
What is effective atomic number of Fe (z = 26) in [Fe(CN)6]4-?
(a) 12
(b) 30
(c) 26
(d) 36
उत्तर
36
In ferrocyanide ion, [Fe(CN)6]4–
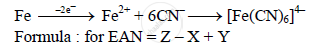
Here, Z = atomic number of iron = 26.
X = number of electrons lost due to oxidation of Fe to Fe2+ = 2.
Y = number of electrons donated by 6CN– = 6 x 2 = 12.
∴ EAN = 26 – 2 + 12 = 36
APPEARS IN
संबंधित प्रश्न
Write down the electronic configuration of Cr3+.
Write down the electronic configuration of Pm3+.
Write down the electronic configuration of Cu+.
Write down the electronic configuration of Ce4+.
Write down the electronic configuration of Mn2+.
Write the different oxidation states of manganese.
Account for the following:
Mn2+ is more stable than Fe2+ towards oxidation to +3 state.
Name the elements of 3d transition series that show maximum number of oxidation states. Why does this happen?
Which transition metal of 3d series has positive E° (M2+/M) value and why?
Out of Cr3+ and Mn3+, which is a stronger oxidising agent and why?
Which among the following elements does not belong to the first transition series?
Which among the following is not considered as a part of transition elements?
Electronic configuration of Cu3+ is ____________.
The electronic configuration of \[\ce{Cu(II)}\] is 3d9 whereas that of \[\ce{Cu(I)}\] is 3d10. Which of the following is correct?
Which of the following compounds is expected to be colored?
Explain the observation, at the end of each period, there is a slight increase in the atomic radius of d-block elements.
Name the element of 3d series which exhibits the largest number of oxidation states. Give reason.
Give reason for the following statements:
Scandium (Z = 21) is a transition element but Zn (Z = 30) is not.
Define transition metals.
Assertion (A) : Copper is a non-transition element.
Reason (R) : Copper has completely filled d-orbitals in its ground state.
Match the properties with the elements of 3d series:
| (i) | lowest enthalpy of atomisation | (p) | Sc |
| (ii) | shows maximum number of oxidation states | (q) | Mn |
| (iii) | transition metal that does not form coloured compounds |
(r) | Zn |
| (s) | Ti |
Write down the electronic configuration of:
\[\ce{CO^2+}\]
