Advertisements
Advertisements
प्रश्न
What kind of particles will be found in a liquid compound which is a non-electrolyte?
उत्तर
Non-electrolyte contains molecules.
APPEARS IN
संबंधित प्रश्न
Name the gas that is produced in each of the given case :
At the anode during the electrolysis of acidified water.
Classify the following substance:
Ammonium chloride
Explain how electrolysis is an example of Redox reaction.
The following is a sketch of an electrolytic cell used in the extraction of aluminium:

- What is the substance of which the electrodes A and B are made?
- At which electrode (A or B) is aluminium formed?
- What are the two aluminium compounds in the electrolyte C?
- Why is it necessary for electrode B to be continuously replaced?
A metal article is to be electroplated with silver. The electrolyte selected is sodium argentocyanide.
- What kind of salt is sodium argentocyanide?
- Why is it preferred to silver nitrate as an electrolyte?
- State one condition to ensure that the deposit is smooth, firm and long lasting.
- Write the reaction taking place at the cathode.
- Write the reaction taking place at the anode.
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
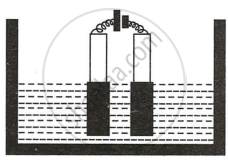
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
Give reasons why in the electroplating of an article with silver, the electrolyte sodium argentocynide solution is preferred over silver nitrate solution.
Copper sulphate solution is electrolysed using copper electrodes.
Which electrode [cathode or anode] is the oxidizing electrode? Why?
Classify the following substance:
Ammonium hydroxide
Classify the following substance:
Dilute hydrochloric acid
