Advertisements
Advertisements
प्रश्न
What is meant by the metal reactivity series ? State its importance, (any two points).
उत्तर
A list in which the metals are arranged in the decreasing order of their chemical reactivity is called the metal reactivity series.

Special features of the activity series:
- The ease with which a metal in solution loses electron(s) and forms a positive ion decreases down the series, i.e. from potassium to gold.
- Hydrogen is included in the activity series because, like metals do, it too loses an electron and becomes positively charged (H+) in most chemical reactions.
- The series facilitates the comparative study of metals in terms of the degree of their reactivity.
- The compounds of the metals (oxides, carbonates, nitrates and hydroxides) too can be easily compared.
APPEARS IN
संबंधित प्रश्न
How will you obtain Silver chloride from silver nitrate.
Also give balanced equations for the reactions
Divide the metals Cu, Zn, Ca, Mg, Fe, Na, Li into three groups, namely reactive metals, moderately reactive metals and less reactive metals.
Select the correct answer for the statement given below:
A neutral oxide which does not react with an acid or a base to give salt and water.
In preparation of Aqua regia hydrochloric acid and _______ acid are mixed.
Observe the following diagram and identify the type of reaction and write observation.
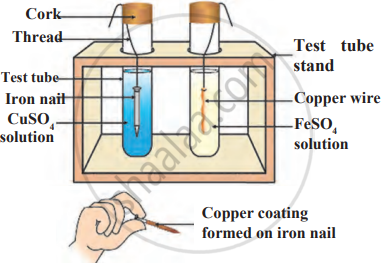
Which of the following is the correct arrangement of the given metals in ascending order of their reactivity?
Zinc, Iron, Magnesium, Sodium
Which among the following statements is incorrect for magnesium metal?
Which among the following alloys contain mercury as one of its constituents?
Of the three metals X, Y and Z. X reacts with cold water, Y with hot water and Z with steam only. Identify X, Y and Z and also arrange them in order of increasing reactivity.
Three metal samples of magnesium, aluminium and iron were taken and rubbed with sandpaper. These samples were then put separately in test tubes containing dilute hydrochloric acid. Thermometers were also suspended in each test tube so that their bulbs dipped in the acid. The rate of formation of bubbles was observed. The above activity was repeated with dilute nitric acid and the observations were recorded.
Answer the following questions:
(i) When the activity was done with dilute hydrochloric acid, then in which one of the test tubes was the rate of formation of bubbles the fastest and the thermometer showed the highest temperature?
(ii) Which metal did not react with dilute hydrochloric acid? Give reason.
(iii) Why is hydrogen gas not evolved when a metal reacts with dilute nitric acid? Name the ultimate products formed in the reaction.
OR
Name the type of reaction on the basis of which the reactivity of metals is decided. You have two metals X and Y. How would you decide which is more reactive than the other?
