Advertisements
Advertisements
प्रश्न
In preparation of Aqua regia hydrochloric acid and _______ acid are mixed.
पर्याय
sulphuric acid
nitric acid
carbonic acid
phosphoric acid
उत्तर
In preparation of Aqua regia hydrochloric acid and nitric acid acid are mixed.
APPEARS IN
संबंधित प्रश्न
What do you observe when a few pieces of iron are dropped in a blue solution of copper sulphate?
What do you observe when ferrous sulphate solution is added to an aqueous solution of sodium hydroxide.
How will you obtain Silver chloride from silver nitrate.
Also give balanced equations for the reactions
Write chemical equation for the event.
Aluminium came in contact with air.
Write chemical equation for the event.
Iron filings are dropped in aqueous solution of copper sulphate.
Write the chemical equation for the event.
A reaction was brought about between ferric oxide and aluminum.
Write a chemical equation for the following event.
Zinc oxide is dissolved in dilute hydrochloric acid.
With reference to Water explain with suitable examples of how the reactivity of the metals could be differentiated.
Select the correct answer for the statement given below:
A neutral oxide which does not react with an acid or a base to give salt and water.
Explain the following reaction with the balanced equation.
Sodium burns in air
Explain the following reaction with the balanced equation.
Reaction of aluminium with oxygen
Explain the following reaction with the balanced equation.
Sulphur burns in air
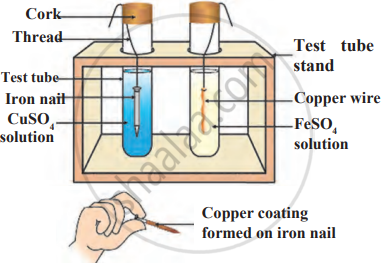
Observe the following diagram and identify the type of reaction and write observation.
Which among the following statements is incorrect for magnesium metal?
Compound X and aluminium are used to join railway tracks.
- Identify the compound X
- Name the reaction
- Write down its reaction.
An element A reacts with water to form a compound B which is used in white washing. The compound B on heating forms an oxide C which on treatment with water gives back B. Identify A, B and C and give the reactions involved.
A metal M does not liberate hydrogen from acids but reacts with oxygen to give a black colour product. Identify M and black coloured product and also explain the reaction of M with oxygen.
Metal ‘A’ has electronic configuration 2, 8, 1 and metal ‘B’ has electronic configuration 2, 8, 8, 2. Out of these, which metal is more reactive? Write the reaction of this metal with dilute HCl acid.
