Advertisements
Advertisements
प्रश्न
Explain the following reaction with the balanced equation.
Reaction of aluminium with oxygen
उत्तर
Aluminium combines with oxygen to form aluminium oxide.
\[\ce{\underset{\text{Aluminium}}{4Al_{(s)}} + \underset{\text{Oxygen}}{3O_{2(g)}} -> \underset{\text{Aluminium oxide}}{2Al2O_{3(s)}}}\]
APPEARS IN
संबंधित प्रश्न
What do you observe when a few pieces of iron are dropped in a blue solution of copper sulphate?
What do you observe when silver nitrate is added to a solution of sodium chloride?
What do you observe when ferrous sulphate solution is added to an aqueous solution of sodium hydroxide.
How will you obtain Silver chloride from silver nitrate.
Also give balanced equations for the reactions
Write chemical equation for the event.
Aluminium came in contact with air.
Write a chemical equation for the following event.
Zinc oxide is dissolved in dilute hydrochloric acid.
With reference to Acid explain with a suitable example of how the reactivity of the metals could be differentiated.
Select the correct answer for the statement given below:
A neutral oxide which does not react with an acid or a base to give salt and water.
Complete the statement by filling in the blank with the correct word:
The metal which reacts with steam and the reaction is reversible is ________.
In preparation of Aqua regia hydrochloric acid and _______ acid are mixed.
Explain the following reaction with the balanced equation.
Sodium burns in air
Explain the following reaction with the balanced equation.
Magnesium reacts with dil HCl
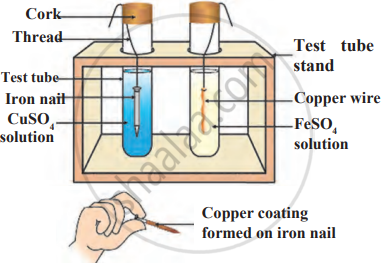
Observe the following diagram and identify the type of reaction and write observation.
Which of the following is the correct arrangement of the given metals in ascending order of their reactivity?
Zinc, Iron, Magnesium, Sodium
Which among the following alloys contain mercury as one of its constituents?
An element A reacts with water to form a compound B which is used in white washing. The compound B on heating forms an oxide C which on treatment with water gives back B. Identify A, B and C and give the reactions involved.
A solution of CuSO4 was kept in an iron pot. After few days the iron pot was found to have a number of holes in it. Explain the reason in terms of reactivity. Write the equation of the reaction involved.
Explain the following
- Reactivity of Al decreases if it is dipped in HNO3
- Carbon cannot reduce the oxides of Na or Mg
- NaCl is not a conductor of electricity in solid state whereas it does conduct electricity in aqueous solution as well as in molten state
- Iron articles are galvanised.
- Metals like Na, K, Ca and Mg are never found in their free state in nature.
