Advertisements
Advertisements
प्रश्न
With reference to Acid explain with a suitable example of how the reactivity of the metals could be differentiated.
उत्तर
Action of metals with acids: As reactivity decreases from top to bottom in metal activity series.
K and Na react with dil. HC1 and dil. H2SO4 explosively to produce H2↑
\[\ce{2Na + 2HCl->2NaCl + H2}\]
Ca, Mg, Al, Zn, and iron react less vigorously with decreasing vigour with dil. H2SO4 or dil. HC1 to produce H2↑
\[\ce{Ca + H2SO->CaSO4 + H2}\]
Metals below hydrogen do not react with dil. acid and do not displace H2 from it.
APPEARS IN
संबंधित प्रश्न
Fill in the blank
When a piece of copper is added to silver nitrate solution, it turns ............in colour.
How will you obtain Magnesium oxide from magnesium.
Also give balanced equations for the reactions
How will you obtain Silver chloride from silver nitrate.
Also give balanced equations for the reactions
Write a chemical equation for the following event.
Zinc oxide is dissolved in dilute hydrochloric acid.
Explain the following reaction with the balanced equation.
Sodium burns in air
Explain the following reaction with the balanced equation.
Sulphur burns in air
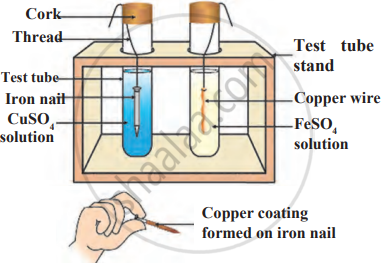
Observe the following diagram and identify the type of reaction and write observation.
Which among the following statements is incorrect for magnesium metal?
A metal M does not liberate hydrogen from acids but reacts with oxygen to give a black colour product. Identify M and black coloured product and also explain the reaction of M with oxygen.
Arrange the following as per the instruction given in the bracket:
Al, K, Mg, Ca (decreasing order of its reactivity)
