Advertisements
Advertisements
प्रश्न
With reference to Acid explain with a suitable example of how the reactivity of the metals could be differentiated.
उत्तर
Action of metals with acids: As reactivity decreases from top to bottom in metal activity series.
K and Na react with dil. HC1 and dil. H2SO4 explosively to produce H2↑
\[\ce{2Na + 2HCl->2NaCl + H2}\]
Ca, Mg, Al, Zn, and iron react less vigorously with decreasing vigour with dil. H2SO4 or dil. HC1 to produce H2↑
\[\ce{Ca + H2SO->CaSO4 + H2}\]
Metals below hydrogen do not react with dil. acid and do not displace H2 from it.
APPEARS IN
संबंधित प्रश्न
What do you observe when dilute sulphuric acid is added to granulated zinc?
What do you observe when ferrous sulphate solution is added to an aqueous solution of sodium hydroxide.
Fill in the blank
When a piece of copper is added to silver nitrate solution, it turns ............in colour.
What do you observe when Magnesium ribbon is burnt in oxygen.
What is meant by the metal reactivity series ? State its importance, (any two points).
Complete the statement by filling in the blank with the correct word:
The metal which reacts with steam and the reaction is reversible is ________.
Explain the following reaction with the balanced equation.
Reaction of aluminium with oxygen
Explain the following reaction with the balanced equation.
Sulphur burns in air
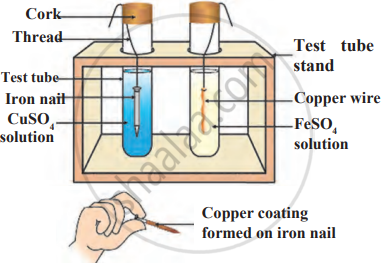
Observe the following diagram and identify the type of reaction and write observation.
A metal that exists as a liquid at room temperature is obtained by heating its sulphide in the presence of air. Identify the metal and its ore and give the reaction involved.
