Advertisements
Advertisements
प्रश्न
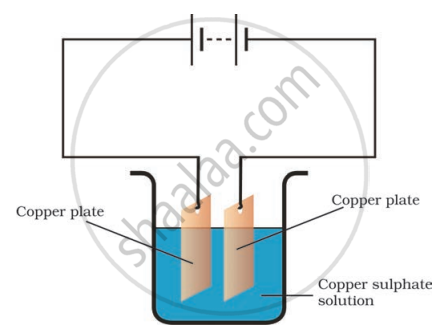
When electric current is passed through a conducting solution, there is a change of colour of the solution. This indicates
पर्याय
the chemical effect of current
the heating effect of current
the magnetic effect of current
the lightning effect of current
उत्तर
the chemical effect of current
Explanation -
The passage of an electric current through a conducting solution causes chemical reactions. As a result, change of colour of solution occurs. This indicates the chemical effect of current.
APPEARS IN
संबंधित प्रश्न
If you pass current through copper sulphate solution, copper gets deposited on the plate connected to the ______ terminal of the battery.
Prepare a list of objects around you that are electroplated.
Boojho’s uncle has set up an electroplating factory near his village. He should dispose off, the waste of the factory
The object to be electroplated is taken as ______ electrode.
In the circuit given in the figure,
Boojho observed that copper is deposited on the electrode connected to the negative terminal of the battery.
Paheli tried to repeat the same experiment. But she could find only one copper plate. Therefore, she took a carbon rod as negative electrode. Will copper be still deposited on the carbon rod? Explain your answer.
You are provided with a magnetic compass, an empty matchbox, a battery of two cells and connecting wires. Using these objects, how will you make a tester for testing an electric circuit? Draw the necessary circuit diagram and explain.
A chemical reaction happens when electricity passes through various conducting liquids.
Liquids that conduct electricity are the solutions of ______.
______ is used in the extraction and purification of metals.
Assertion: Parts of cars and bicycles are made iron with chromium coating.
Reason: Chromium does not corrode and resist scratches.
