Advertisements
Advertisements
प्रश्न
When water is cooled to a temperature x, it gets converted into ice at temperature x by a process called P. And when ice at temperature x is warmed, it gets reconverted into water at the same temperature x in a process called Q.
- What is the value of temperature x in Kelvin ?
- What is the process P known as ?
- What is the name of energy released during process P ?
- What is the process Q known as ?
- What is the name of energy absorbed during process Q ?
उत्तर
- 273K
- The name of the process is freezing.
- Latent heat of freezing
- Melting
- Latent heat of fusion
APPEARS IN
संबंधित प्रश्न
Suggest a method to liquefy atmospheric gases.
Give two reasons to justify water at room temperature is a liquid.
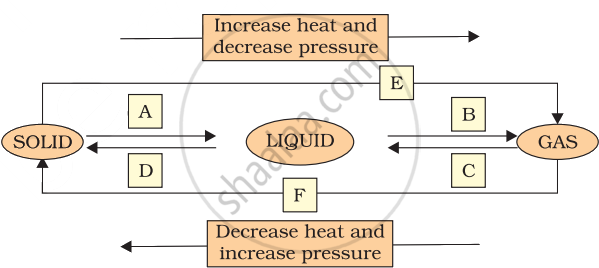
Name A, B, C, D, E and F in the following diagram showing change in its state.
What do you observe when :
ime is dissolved in water.
Does the substance absorb or liberate any heat energy during the change of phase?
Fill in the following blank with suitable words :
Gases can be liquefied by applying ....................... and lowering .....................
Draw a labelled diagram of the experimental set-up to study the latent heat of vaporisation of water.
Differentiate between: Melting and boiling
Give reasons:
How is interconversion of states of matter different from chemical reaction?
The process by which a solid changes into a liquid is called
