Advertisements
Advertisements
प्रश्न
Which of the following process/processes release heat ?
- condensation
- vaporisation
- freezing
- melting
पर्याय
only (i)
only (iv)
(i) and (iii)
(ii) and (iv)
उत्तर
(i) and (iii)
Both condensation and freezing release heat that lead to decrease in kinetic energy of molecules. When the substance gets cool enough its particles become slower or stop their movement and change their phase into liquid and solid on condensing and freezing respectively.
APPEARS IN
संबंधित प्रश्न
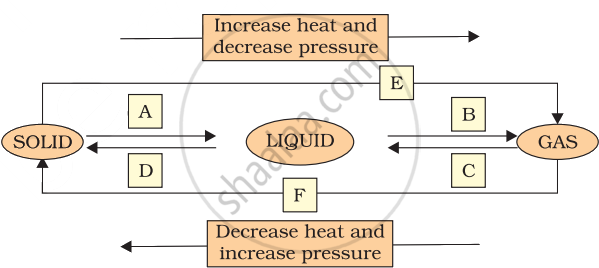
Name A, B, C, D, E and F in the following diagram showing change in its state.
Is there any change in temperature during the change of phase?
What is the name of the process due to which dry ice changes into carbon dioxide gas ?
State whether the following statement is true or false :
Solid carbon dioxide is stored under low pressure.
What is the chemical name of dry ice ?
Explain why, ice at 0° C is more effective in cooling than water at the same temperature.
Which contains more heat, 1 kg of water at 100° C or 1 kg of steam at 100° C ? Give reason for your answer.
Why does a desert cooler cool better on a hot, dry day ?
When water is cooled to a temperature x, it gets converted into ice at temperature x by a process called P. And when ice at temperature x is warmed, it gets reconverted into water at the same temperature x in a process called Q.
- What is the value of temperature x in Kelvin ?
- What is the process P known as ?
- What is the name of energy released during process P ?
- What is the process Q known as ?
- What is the name of energy absorbed during process Q ?
When water is heated to a temperature x, it gets converted into steam at temperature x by a process called R. And when steam at temperature x is cooled, it gets reconverted into water at the same temperature x by a process called S.
- How much is the value of x in Kelvin ?
- What is the process R called ?
- What is the name of the energy absorbed during the process R ?
- What is process S known as ?
- What is the name of energy released during the process S known as ?\\
