Advertisements
Advertisements
प्रश्न
Which of the following equation depicts reducing nature of \[\ce{H2O2}\]?
पर्याय
\[\ce{2[Fe(CN)6]^{4-} + 2H^{+} + H2O2 -> 2[Fe(CN)6]^{3-} + 2H2O}\]
\[\ce{I2 + H2O2 + 2OH^{-} -> 2I^{-} + 2H2O + O2}\]
\[\ce{Mn^{2+} + H2O2 -> Mn^{4+} + 2OH-}\]
\[\ce{PbS + 4H2O2 -> PbSO4 + 4H2O}\]
उत्तर
\[\ce{I2 + H2O2 + 2OH^{-} -> 2I^{-} + 2H2O + O2}\]
Explanation:
As iodine is reduced from 0 to –1.
APPEARS IN
संबंधित प्रश्न
Arrange the following:
CaH2, BeH2 and TiH2 in order of increasing electrical conductance.
Why is it said that-
There is no alternative to water for cleaning purposes.
Explain the picture in your own words.
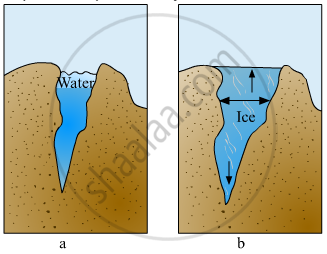
Why is the density of seawater more than that of rain water?
Define the following.
Boiling point
Which of the following equations depict the oxidising nature of \[\ce{H2O2}\]?
Explain why \[\ce{HCl}\] is a gas and \[\ce{HF}\] is a liquid.
Write two reactions to explain amphoteric nature of water.
Which of the following ion is responsible for the temporary hardness of water?
The density of gold is 19 g/cm3. If 1.9 × 10−4 g of gold is dispersed in one litre of water to give a sol having spherical gold particles of radius 10 nm, the number of gold particles per mm3 of the sol will be ______ × 106.
