Advertisements
Advertisements
प्रश्न
Which of the following equations depict the oxidising nature of \[\ce{H2O2}\]?
पर्याय
\[\ce{2MnO^{-}4 + 6H^{+} + 5H2O2 -> 2Mn^{2+} + 8H2O + 5O2}\]
\[\ce{2Fe^{3+} + 2H^{+} + H2O2 -> 2Fe^{2+} + 2H2O + O2}\]
\[\ce{2I^{-} + 2H^{+} + H2O2 -> I2 + 2H2O}\]
\[\ce{KIO4 + H2O2 -> KIO3 + H2O + O2}\]
उत्तर
\[\ce{2I^{-} + 2H^{+} + H2O2 -> I2 + 2H2O}\]
Explanation:
\[\ce{2\overset{-1}{I^{-}} + 2\overset{+1}{H^{+}} + \overset{+1}{H2}\overset{-1}{O2} -> \overset{0}{I2} + 2\overset{+1}{H2}\overset{-2}{O}}\]
\[\ce{I-}\] ions are oxidized to \[\ce{I2}\] (increase in O.N. from –1 to 0), Hence, \[\ce{H2O2}\] acts as an oxidizing agent.
APPEARS IN
संबंधित प्रश्न
Describe the usefulness of water in biosphere and biological systems.
Explain the picture in your own words.
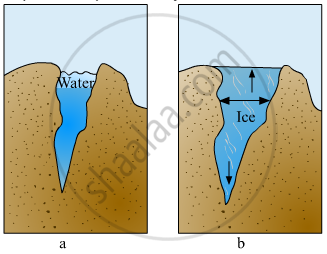
Why should a glass bottle completely filled with water never be kept in a freezer?
Define the following.
Freezing point
Define the following.
Boiling point
Which of the following equation depicts reducing nature of \[\ce{H2O2}\]?
If same mass of liquid water and a piece of ice is taken, then why is the density of ice less than that of liquid water?
Write two reactions to explain amphoteric nature of water.
Pure water boils at ______ °C at one atmospheric pressure.
______ has the highest latent heat of fusion.
