Advertisements
Advertisements
Question
Which of the following equations depict the oxidising nature of \[\ce{H2O2}\]?
Options
\[\ce{2MnO^{-}4 + 6H^{+} + 5H2O2 -> 2Mn^{2+} + 8H2O + 5O2}\]
\[\ce{2Fe^{3+} + 2H^{+} + H2O2 -> 2Fe^{2+} + 2H2O + O2}\]
\[\ce{2I^{-} + 2H^{+} + H2O2 -> I2 + 2H2O}\]
\[\ce{KIO4 + H2O2 -> KIO3 + H2O + O2}\]
Solution
\[\ce{2I^{-} + 2H^{+} + H2O2 -> I2 + 2H2O}\]
Explanation:
\[\ce{2\overset{-1}{I^{-}} + 2\overset{+1}{H^{+}} + \overset{+1}{H2}\overset{-1}{O2} -> \overset{0}{I2} + 2\overset{+1}{H2}\overset{-2}{O}}\]
\[\ce{I-}\] ions are oxidized to \[\ce{I2}\] (increase in O.N. from –1 to 0), Hence, \[\ce{H2O2}\] acts as an oxidizing agent.
APPEARS IN
RELATED QUESTIONS
What properties of water make it useful as a solvent? What types of compound can it (i) dissolve, and (ii) hydrolyse?
Why is it said that-
There is no alternative to water for cleaning purposes.
Explain the picture in your own words.
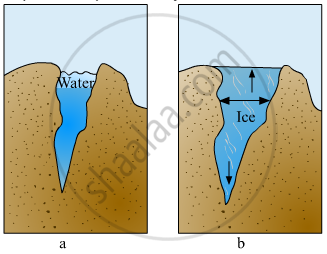
Define the following.
Freezing point
Write two reactions to explain amphoteric nature of water.
The boiling point of water increases with ______ in pressure.
Freezing of water will cause an ______ is the volume.
______ has the highest latent heat of fusion.
One gram of water requires ______ of heat to raise its temperature by l°C.
What are the physical properties of pure water?
