Advertisements
Advertisements
Question
Define the following.
Freezing point
Solution
The temperature at which a liquid turns into solid when cooled is known as the freezing point. The freezing point of water is 0°C.
APPEARS IN
RELATED QUESTIONS
Arrange the following:
CaH2, BeH2 and TiH2 in order of increasing electrical conductance.
Explain the picture in your own words.
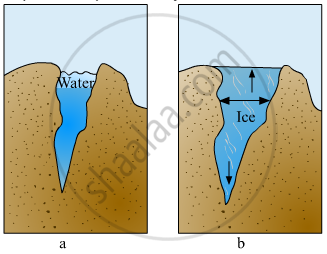
Why is the density of seawater more than that of rain water?
Define the following.
Boiling point
Which of the following equations depict the oxidising nature of \[\ce{H2O2}\]?
Some of the properties of water are described below. Which of them is/are not correct?
(i) Water is known to be a universal solvent.
(ii) Hydrogen bonding is present to a large extent in liquid water.
(iii) There is no hydrogen bonding in the frozen state of water.
(iv) Frozen water is heavier than liquid water.
Give reasons: Ice floats on water.
Write two reactions to explain amphoteric nature of water.
Water is circulated around the car engine using the ______ pump and the heat is absorbed.
What are the physical properties of pure water?
