Advertisements
Advertisements
प्रश्न
Which of the following is an amorphous solid?
पर्याय
Graphite (C)
Quartz glass (SiO2)
Chrome alum
Silicon carbide (SiC)
उत्तर
Quartz glass (SiO2)
Explanation:
| Crystalline Silica (Quartz) | Amorphous Silica (Glass) | |
| 1. | Crystalline in nature. Also called quartz. | Light white powder. |
| 2. | All four corners of \[\ce{SiO4}\] tetrahedron are shared by others to give a network solid. | The \[\ce{sio4}\] tetrahedron are randomly joined giving rise to polymeric chains, sheets or three- dimensional units |
| 3. | High and sharp melting point (1710°C) | Does not have sharp melting point. On heating softens gradually to liquid. |
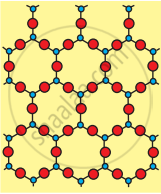
(a) Crystalline (Quartz)
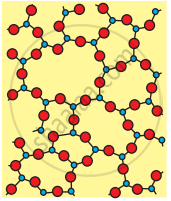
(b) Amorphous (Quartz glass)
APPEARS IN
संबंधित प्रश्न
The relation a ≠ b ≠ c and α ≠ β ≠ γ represents which crystal system?
(A) Orthorhombic
(B) Tetragonal
(C) Triclinic
(D) Monoclinic
Why is glass considered a super cooled liquid?
Define the term 'amorphous'. Give a few examples of amorphous solids.
Why does sodium chloride on heating with sodium vapours acquire yellow colour?
Most crystals show good cleavage because their atoms, ions or molecules are ____________.
Which of the following statements about amorphous solids is incorrect?
Why some glass objects from ancient civilisations are found to become milky in appearance?
Which of the following amorphous solid is used as photovoltaic material for conversion of sunlight into electricity?
Which of the following is true about the value of refractive index of quartz glass?
Amorphous solid can also be called:
(i) pseudo solids
(ii) true solids
(iii) supercooled liquids
(iv) supercooled solids
