Advertisements
Advertisements
प्रश्न
Which of the following species can act as the strongest base?
पर्याय
ΘOH
ΘOR
ΘOC6H5
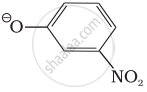
उत्तर
ΘOR
Explanation:
Weakest acid has the strongest conjugate base. Since R-OH is the weakest acid, therefore, RO is the strongest base.
APPEARS IN
संबंधित प्रश्न
While separating a mixture of ortho and para nitrophenols by steam distillation, name the isomer which will be steam volatile. Give reason.
Picric acid is ____________.
When phenol is treated with excess bromine water, it gives:
The electrophile involved in Reimer-Tiemann reaction of phenol with CHCl3 in presence of NaOH:

Which of the following reactions is used to prepare salicylaldehyde?
Which of the following compounds is aromatic alcohol?
| (A) |  |
| (B) | 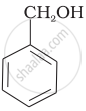 |
| (C) | 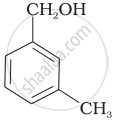 |
| (D) | 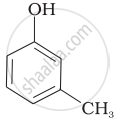 |
Which of the following are benzylic alcohols?
(i) \[\ce{C6H5 - CH2 - CH2OH}\]
(ii) \[\ce{C6H5 - CH2OH}\]
(iii) \[\begin{array}{cc}
\ce{C6H5 - CH - OH}\\
\phantom{}|\phantom{.}\\
\phantom{..}\ce{CH3}\phantom{}
\end{array}\]
(iv) \[\begin{array}{cc}
\ce{C6H5 - CH2 - CH - OH}\\
\phantom{.......}|\phantom{}\\
\phantom{.........}\ce{CH3}\phantom{}
\end{array}\]
Convert the following:
Phenol to N-phenylethanamide.
Write the equations for the following reaction:
Phenol is treated with chloroform in the presence of NaOH
