Advertisements
Advertisements
प्रश्न
Which of the following are benzylic alcohols?
(i) \[\ce{C6H5 - CH2 - CH2OH}\]
(ii) \[\ce{C6H5 - CH2OH}\]
(iii) \[\begin{array}{cc}
\ce{C6H5 - CH - OH}\\
\phantom{}|\phantom{.}\\
\phantom{..}\ce{CH3}\phantom{}
\end{array}\]
(iv) \[\begin{array}{cc}
\ce{C6H5 - CH2 - CH - OH}\\
\phantom{.......}|\phantom{}\\
\phantom{.........}\ce{CH3}\phantom{}
\end{array}\]
उत्तर
(ii) \[\ce{C6H5 - CH2OH}\]
(iii) \[\begin{array}{cc}
\ce{C6H5 - CH - OH}\\
\phantom{}|\phantom{.}\\
\phantom{..}\ce{CH3}\phantom{}
\end{array}\]
Explanation:
In these alcohols, the –OH group is attached to a sp3- hybridised carbon atom next to an aromatic ring. For example,
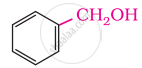
Primary
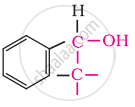
Secondary
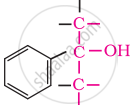
Tertiary
Allylic and benezlic alcohols may be primary, secondary or tertiary.
APPEARS IN
संबंधित प्रश्न
Write the main products when
2, 4, 6-trinitrochlorobenzene is subjected to hydrolysis
Write the main product(s) in each of the following reactions:
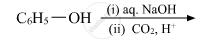
Picric acid is ____________.
When phenol is treated with excess bromine water, it gives:
On distilling phenol with Zn dust, one gets:
Which of the following species can act as the strongest base?
Which of the following is not aromatic?
For the pair phenol and cyclohexanol, answer the following:
Give one chemical test to distinguish between the two.
How can phenol be prepared from anisole? Give reaction.
Convert the following by giving a chemical equation:
Phenol to salicylaldehyde.
