Advertisements
Advertisements
प्रश्न
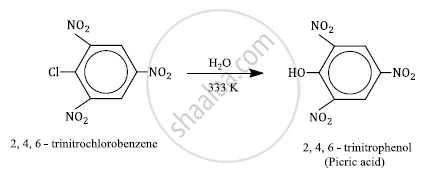
Write the main products when
2, 4, 6-trinitrochlorobenzene is subjected to hydrolysis
उत्तर
2, 4, 6-trinitrochlorobenzene under mild hydrolysis conditions (H2O/323 K) gives 2, 4, 6-trinitrophenol or picric acid.
APPEARS IN
संबंधित प्रश्न
Give simple chemical tests to distinguish between the following pairs of compounds: Benzoic acid and Phenol
Give the equation of the following reaction:
Bromine in CS2 with phenol.
When phenol is treated with excess bromine water, it gives:
The electrophile involved in Reimer-Tiemann reaction of phenol with CHCl3 in presence of NaOH:
Which of the following reactions is used to prepare salicylaldehyde?
Phenol does not undergo nucleophilic substitution reaction easily due to ______.
Out of o-nitrophenol and p-nitrophenol, which is more volatile? Explain.
Write the equations for the following reaction:
Phenol is treated with chloroform in the presence of NaOH
For the pair phenol and cyclohexanol, answer the following:
Give one chemical test to distinguish between the two.
Convert the following by giving a chemical equation:
Phenol to salicylaldehyde.
