Advertisements
Advertisements
प्रश्न
Give simple chemical tests to distinguish between the following pairs of compounds: Benzoic acid and Phenol
उत्तर १
(ii) Distinguish test between Benzoic acid and Phenol:
NaHCO3 Test: When Benzoic acid reacts with NaHCO3, brisk effervescence of CO2 gas evolved.

Phenol does not give this test.
C6H5OH + NaHCO3 → No Reaction.
उत्तर २
Benzoic acid and Phenol : Benzoic acid and phenol can be distinguish by FeCl3 tests. Both reacts with FeCl3 to give different colours. Phenol reacts with FeCl3 to give violet coloured precipitate while benzoic acid give buff coloured precipitate.

उत्तर ३
When treated with sodium bicarbonate, benzoic acid gives brisk effervescence due to evolution of carbon dioxide gas. No reaction takes place when phenol is treated with NaHCO3.
C6H5COOH + NaHCO3 → C6H5COONa + H2O + CO2
APPEARS IN
संबंधित प्रश्न
Write the equation involved in the following reaction:
Reimer-Tiemann reaction
Picric acid is ____________.
In the reaction of phenol with CHCl3 and aqueous NaOH at 343 K, the electrophile attacking the ring is:
Which of the following species can act as the strongest base?
Which of the following compounds is aromatic alcohol?
| (A) |  |
| (B) | 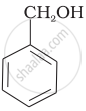 |
| (C) | 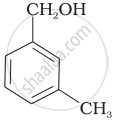 |
| (D) | 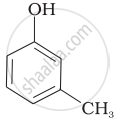 |
Which of the following are used to convert \[\ce{RCHO}\] into \[\ce{RCH2OH}\]?
(i) \[\ce{H2/Pd}\]
(ii) \[\ce{LiAlH4}\]
(iii) \[\ce{NaBH4}\]
(iv) Reaction with \[\ce{RMgX}\] followed by hydrolysis
Which of the following are benzylic alcohols?
(i) \[\ce{C6H5 - CH2 - CH2OH}\]
(ii) \[\ce{C6H5 - CH2OH}\]
(iii) \[\begin{array}{cc}
\ce{C6H5 - CH - OH}\\
\phantom{}|\phantom{.}\\
\phantom{..}\ce{CH3}\phantom{}
\end{array}\]
(iv) \[\begin{array}{cc}
\ce{C6H5 - CH2 - CH - OH}\\
\phantom{.......}|\phantom{}\\
\phantom{.........}\ce{CH3}\phantom{}
\end{array}\]
Convert the following:
Phenol to N-phenylethanamide.
Attacking species in nitration of benzene in presence of fuming HNO3 is
Convert the following by giving a chemical equation:
Phenol to salicylaldehyde.
